Abstract
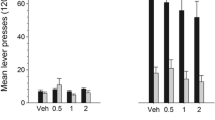
The non-selective opioid antagonist naltrexone and the partial agonist buprenorphine have been reported to reduce cocaine self-administration (SA) and relapse in both humans and rhesus monkeys. Data suggesting an involvement ofδ-opioid receptors in modulating the conditioned rewarding effects of cocaine were also recently presented. In view of such findings, the present SA and place conditioning studies were conducted to examine the influence of the selectiveδ-opioid receptor antagonist naltrindole upon the rewarding effects of cocaine. Sprague-Dawley rats were trained to self-administer cocaine (1.0 mg/kg per infusion) on an FR2 schedule of reinforcement. Dose-response and antagonist testing commenced once stable rates of cocaine SA were achieved. For antagonist testing, rats received naltrindole (0.03–10.0 mg/kg, IP) 30 min prior to the start of 2-h SA sessions. SA behavior in response to cocaine delivery (0.25 and 1.0 mg/kg per infusion) was then determined. Naltrindole in doses of 0.03–3.0 mg/kg did not alter the number of cocaine infusions taken by the rats. A higher dose of naltrindole (10.0 mg/kg), which markedly depressed locomotor activity, resulted in a 16% reduction of cocaine (0.25 mg/kg per infusion) SA behavior. When SA sessions were terminated and naltrindole (1.0 mg/kg) was administered repeatedly for 3 days, no alterations in the re-acquisition of cocaine SA were seen. Place conditioning studies also failed to find an effect of naltrindole (0.1–3.0 mg/kg) on cocaine (10 mg/kg) — induced conditioned place preferences. Naltrindole, by itself, did not induce significant place conditioning. These data fail to indicate a role ofδ-opioid receptors in modulating either the positive reinforcing or conditioned rewarding effects of cocaine. Furthermore, they suggest that the therapeutic actions of naloxone, naltrexone and buprenorphine on cocaine SA behavior may not result from the specific blockade ofδ-opioid receptors.
Similar content being viewed by others
References
Bain GT, Kornetsky C (1987) Naloxone attenuation of the effect of cocaine on rewarding brain stimulation. Life Sci 40:1119–1125
Bals-Kubik R, Herz A, Shippenberg TS (1989) Evidence that the aversive effects of opioid antagonists are centrally mediated. Psychopharmacology 98:203–208
Belcheva MM, Barg J, McHale RJ, Dawn S, Ho MT, Ignatova E, Coscia CJ (1993) Differential down- and up-regulation of rat brain opioid receptor types and subtypes by buprenorphine. Mol Pharmacol 44:173–179
Bilsky EJ, Montegut MJ, Delong CL, Reid LD (1992) Opioidergic modulation of cocaine conditioned place preferences. Life Sci 50:PL85–90
Carroll ME, Lac ST (1992) Effects of buprenorphine on self-administration of cocaine and a nondrug reinforcer in rats. Psychopharmacology 106:439–446
Comer SD, Lac ST, Curtis LK, Carrol ME (1993) Effects of buprenorphine and naltrexone on reinstatement of cocaine-reinforced responding in rats. J Pharmacol Exp Ther 267:1470–1476
Corrigal WA, Coen KM (1991) Opiate antagonists reduce cocaine but not nicotine self-administration. Psychopharmacology 104:167–170
Cowan A, Lewis JW, MacFarlane IR (1977) Agonist and antagonist properties of buprenorphine, a new antinociceptive agent. Br J Pharmacol 60:537–545
Drower EJ, Stapelfeld A, Rafferty MF, De Costa BR, Rice KC, Hammond DL (1991) Selective antagonism by naltrindole of the antinociceptive effects of the delta opioid agonist cyclic DPDPE in the rat. J Pharmacol Exp Ther 259:725–731
Hollt V (1983) Multiple endogenous opioid peptide. Trends Neuroscience 6:24–26
Hurd Y, Herkenham M (1992) Influence of a single injection of cocaine, amphetamine or GBR 12909 on mRNA expression of striatal neuropeptides. Mol Brain Res 16:97–104
Jackson HC, Ripley TL, Nutt DJ (1989) Exploringδ-receptor function using the selective opioid antagonist naltrindole. Neuropharmacology 28:1427–1430
Jasinski DR, Pevnick JS, Griffith D (1978) Human pharmacology and abuse potential of the analgesic buprenorphine. Arch Gen Psychiatry 35:601–616
Johnson RE, Jaffe JH, Fudala PJ (1992) A controlled trial of buprenorphine treatment for opioid dependence. J Am Med Assoc 267:2750–2755
Kitchen I, Kennedy A (1990) Effects ofδ-opioid receptor antagonist naltrindole on opioid induced in vivo release of corticosterone. In: Van Ree JM, Mulder AH, Wiegant VM, Wiemersma-Greidanus TB (eds) New leads in opioid research. Excerpta Medica, Amsterdam, pp 2321–2322
Kitchen I, Pinker SR (1990) Antagonism of swim-stress-induced antinociception by theδ-opioid receptor antagonist naltrindole in adult and young rats. Br J Pharmacol 100:685–688
Koob GF, Le HT, Creese I (1987) The D1 dopamine receptor antagonist SCH 23390 increases coccaine self-administration in the rat Neuroscience Letters 79:315–320
Kosten TR, Kleber HD, Morgan C (1989) Treatment of cocaine abuse with buprenorphine. Biol Psychiatry 26:737–739
Leander JD (1987) Buprenorphine has potent kappa opioid receptor antagonist activity. Neuropharmacology 26:1445–1447
Magnan J, Paterson SJ, Tavani A, Kosterlitz HW (1982) The binding spectrum of narcotic analgesic drugs with different agonist and antagonist properties. Naunyn Schmiedeberg's Arch Pharmacol 319:197–200
Mello NK, Mendelson JH (1980) Buprenorphine suppresses heroin use by heroin addicts. Science 207:657–659
Menkens K, Bilsky EJ, Wild KD, Portoghese PS, Reid LD, Porreca F (1992) Cocaine place preference is blocked by theδ-opioid receptor antagonist, naltrindole. Eur J Pharmacol 219:345–346
Negus SS, Henriksen SJ, Mattox A, Pasternak GW, Portoghese PS, Takemori AE, Weiniger MB, Koob GF (1993) Effect of antagonists selective for mu, delta and kappa opioid receptors on the reinforcing effects of heroin in rats. J Pharmacol Exp Ther 265:1245–1252
Portoghese, PS, Sultana M, Takemori AE (1988) Naltrindole a highly selective and potent non peptide S opioid receptor antagonist Eur J Pharmacol 146:185–186
Reid LD, Hubbell CL, Glaccum MB, Bilsky EJ, Portoghese PS, Porreca F (1993) Naltrindole, an opioid delta receptor antagonist, blocks cocaine-induced facilitation of responding for rewarding brain stimulation. Life Sci 52:PL67–71
Richards ML, Sadee W (1985) Buprenorphine is an antagonist at the k opioid receptor. Pharm Res 2:178–181
Schoffelmeer ANM, De Vries TJ, Hogenboom F, Mulder AH (1993) Mu- and delta-opioid receptors inhibitory linked to dopamine-sensitive adenylate cyclase in rat striatum display a selectivity profile towards endogenous opioid peptides different from that of presynaptic mu, delta and kappa receptors. J Pharmacol Exp Ther 267:205–210
Shippenberg TS, Bals-Kubik R, Herz A (1987) Motivational properties of opioids: evidence that an activation ofδ receptors mediates reinforcement processes. Brain Res 436:234–239
Suzuki T, Mori M, Tsuji M, Misawa M, Nagase H (1994) The role ofδ-opioid receptor subtypes in cocaine- and methamphetamine-induced place preferences. Life Sci 55: PL 339–344
Trujillo KA, Belluzzi JD, Stein L (1991) Naloxone blockade of amphetamine place preference conditioning. Psychopharmacology 104:265–274
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Vries, T.J., Babovic-Vuksanovic, D., Elmer, G. et al. Lack of involvement ofδ-opioid receptors in mediating the rewarding effects of cocaine. Psychopharmacology 120, 442–448 (1995). https://doi.org/10.1007/BF02245816
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02245816