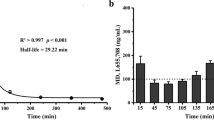
Abstract
Rationale
Compounds selective for the GABAA receptors containing an α5 subunit have been reported to enhance performance in the hippocampally mediated delayed-matching-to-position version of the Morris water maze, in which reduction in the time required to find a hidden platform relative to an initial trial is used as an index of learning and memory.
Objective
In the present study, we have used one such compound, α5IA-II, to examine whether these effects occur during the encoding, consolidation or recall phases of this paradigm.
Methods
α5IA-II was administered in the absence or presence of the benzodiazepine site antagonist flumazenil, so as to limit its action to periods associated with encoding, consolidation and recall. Drug doses and timings of administrations were defined using occupancy data derived from an in vivo [3H]flumazenil binding assay. Similar experiments were carried out to study the memory-disruptive properties of chlordiazepoxide (CDP).
Results
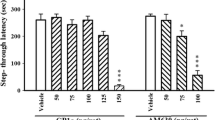
The trial 1 to trial 2 difference was increased when α5IA-II was given before either trial 1 or trial 2, indicating an effect on the encoding and recall phases, respectively, of learning and memory. Conversely, α5IA-II had no effect on performance when given immediately after trial 1, suggesting that it had no effect on the consolidation phase. In contrast to the facilitation of performance produced by the α5-selective inverse agonist α5IA-II given during the encoding and recall but not the consolidation phase, the non-selective agonist CDP impaired performance when given during the encoding and recall phases, whilst having no effect on the consolidation phase.
Conclusions
These data further highlight the cognition-enhancing properties of GABAA α5-selective inverse agonists and define the functional specificity of these effects in terms of encoding and recall processes in the Morris water maze.







Similar content being viewed by others
References
Abel T, Lattal KM (2001) Molecular mechanisms of memory acquisition, consolidation and retrieval. Curr Opin Neurobiol 11:180–187
Argyropoulos SV, Nutt DJ (1999) The use of benzodiazepines in anxiety and other disorders. Eur Neuropsychopharmacol 9(Suppl 6):S391–S392
Atack JR (2005) The benzodiazepine binding site of GABAA receptors as a target for the development of novel anxiolytics. Expert Opin Investig Drugs 14:601–618
Atack JR, Hutson PH, Collinson N, Marshall G, Bentley G, Moyes C, Cook SM, Collins I, Wafford K, McKernan RM, Dawson GR (2005) Anxiogenic properties of an inverse agonist selective for α3 subunit-containing GABAA receptors. Br J Pharmacol 144:357–366
Atack JR, Wafford KA, Tye SJ, Cook SM, Sohal S, Pike A, Sur C, Melillo D, Bristow L, Bromidge F, Ragan I, Kerby J, Street L, Carling R, Castro J-L, Whiting P, Dawson GR, McKernan RM (2006) TPA023 [7-(1,1-dimethylethyl)-6-(2-ethyl-2H-1,2,4-triazol-3-ylmethoxy)-3-(2-fluorophenyl)-1,2,4-triazolo[4,3-b]pyridazine], an agonist selective for α2- and α3-containing GABAA receptors, is a non-sedating anxiolytic in rodents and primates. J Pharmacol Exp Ther 316:410–422
Brown N, Kerby J, Bonnert TP, Whiting PJ, Wafford KA (2002) Pharmacological characterization of a novel cell line expressing human α4β3δ GABAA receptors. Br J Pharmacol 136:965–974
Chambers MS, Atack JR, Carling RW, Collinson N, Cook SM, Dawson GR, Ferris P, Hobbs SC, O’Connor D, Marshall G, Rycroft W, Macleod AM (2004) An orally bioavailable, functionally selective inverse agonist at the benzodiazepine site of GABAA α5 receptors with cognition enhancing properties. J Med Chem 47:5829–5832
Cobain MR (1999) The localization and function of the alpha5 subunit containing GABAA receptors in the rat. PhD thesis, Downing College, Cambridge
Collinson N, Kuenzi F, Jarolimek W, Maubach KA, Cothliff R, Sur C, Smith A, Otu FM, Howell O, Atack JR, McKernan RM, Seabrook GR, Dawson GR, Whiting PW, Rosahl TW (2002) Enhanced learning and memory and altered GABAergic synaptic transmission in mice lacking the α5 subunit of the GABAA receptor. J Neurosci 22:5572–5580
Crestani F, Keist R, Fritschy J-M, Benke D, Vogt K, Prut L, Blüthmann H, Möhler H, Rudolph U (2002) Trace fear conditioning involves hippocampal α5 GABAA receptors. Proc Natl Acad Sci U S A 99:8980–8985
Davis HP, Squire LR (1984) Protein synthesis and memory: a review. Psychol Bull 96:518–559
Dawson GR, Maubach KA, Collinson N, Cobain M, Everitt BJ, MacLeod AM, Choudhury HI, McDonald LM, Pillai G, Rycroft W, Smith AJ, Sternfeld F, Tattersall FD, Wafford KA, Reynolds DS, Seabrook GR, Atack JR (2006) An inverse agonist selective for α5 subunit-containing GABAA receptors enhances cognition. J Pharmacol Exp Ther 316:1335–1345
Dias R, Sheppard WFA, Fradley RL, Garrett EM, Stanley JL, Marshall GR, Goodacre S, Lincoln RJ, Tye SJ, Cook S, Conley R, Hallett D, Wafford KA, Street LJ, Castro JL, Whiting PJ, Rosahl TW, Atack JR, McKernan R, Dawson GR, Reynolds DS (2005) Anxiolytic properties of benzodiazepines are mediated through the GABAA α3 receptor subtype. J Neurosci 25:10682–10688
Dorow R, Horowsli R, Paschelke G, Amin M, Braestrup C (1983) Severe anxiety induced by FG 7142, a β-carboline ligand for benzodiazepine receptors. Lancet 2:98–99
Duka T, Curran HV, Rusted JM, Weingartner HJ (1996a) Perspectives on cognitive psychopharmacology research. Behav Pharmacol 7:401–410
Duka T, Ott H, Rohloff A, Voet B (1996b) The effects of a benzodiazepine receptor antagonist beta-carboline ZK-93426 on scopolamine-induced impairment on attention, memory and psychomotor skills. Psychopharmacology (Berl) 123:361–373
Fritschy JM, Möhler H (1995) GABAA receptor heterogeneity in the adult rat brain: differential regional and cellular distribution of seven major subunits. J Comp Neurol 359:154–194
Ghoneim MM, Mewaldt SP (1975) Effects of diazepam and scopolamine on storage, retrieval and organizational processes in memory. Psychopharmacologia 44:257–262
Ghoneim MM, Mewaldt SP (1990) Benzodiazepines and human memory: a review. Anesthesiology 72:926–938
Ghoneim MM, Hinrichs JV, Mewaldt SP (1984) Dose response analysis of the behavioral effects of diazepam: I. Learning and memory. Psychopharmacology (Berl) 82:291–295
Goelet P, Castellucci VF, Schacher S, Kandel ER (1986) The long and short of long-term memory—a molecular framework. Nature 322:419–422
Hinrichs JV, Ghoneim MM, Mewaldt SP (1984) Diazepam and memory: retrograde facilitation produced by interference reduction. Psychopharmacology (Berl) 84:158–162
Jensen LH, Stephens DN, Sarter M, Petersen EN (1987) Bidirectional effects of β-carbolines and benzodiazepines on cognitive processes. Brain Res Bull 19:359–364
Löw K, Crestani F, Keist R, Benke D, Brünig I, Benson JA, Fritschy JM, Rülicke T, Bluethmann H, Möhler H, Rudolph U (2000) Molecular and neuronal substrate for the selective attenuation of anxiety. Science 290:131–134
Maubach K (2003) GABAA receptor subtype selective cognition enhancers. Curr Drug Target CNS Neurol Disord 2:233–239
McKernan RM, Rosahl TW, Reynolds DS, Sur C, Wafford KA, Atack JR, Farrar S, Myers J, Cook G, Ferris P, Garret L, Bristow L, Marshall G, Macaulay A, Brown N, Howell O, Moore KW, Carling RW, Street LJ, Castro JL, Ragan CI, Dawson GR, Whiting PJ (2000) Sedative but not anxiolytic properties of benzodiazepines are mediated by the GABAA receptor α1 subtype. Nat Neurosci 3:587–592
McNamara RK, Skelton RW (1993) Effects of intracranial infusions of chlordiazepoxide on spatial learning in the Morris water maze. I. Neuroanatomical specificity. Behav Brain Res 59:175–191
McNaughton N, Morris RGM (1987) Chlordiazepoxide, an anxiolytic benzodiazepine impairs place navigation in rats. Behav Brain Res 24:39–46
Milner B, Squire LR, Kandel ER (1998) Cognitive neuroscience and the study of memory. Neuron 20:445–468
Minier F, Sigel E (2004) Techniques: use of concatenated subunits for the study of ligand-gated ion channels. Trends Pharmacol Sci 25:499–503
Morris RG, Garrud P, Rawlins JN, O’Keefe J (1982) Place navigation impaired in rats with hippocampal lesions. Nature 297:681–683
Nadel L, Land C (2000) Memory traces revisited. Nat Rev Neurosci 1:209–212
Nadel L, Moscovitch M (1997) Memory consolidation, retrograde amnesia and the hippocampal complex. Curr Opin Neurobiol 7:217–227
Nadel L, Moscovitch M (1998) Hippocampal contributions to cortical plasticity. Neuropharmacology 37:431–439
Nader K, Schafe GE, LeDoux JE (2000) Fear memories require protein synthesis in the amygdala for reconsolidation after retrieval. Nature 406:722–726
Petersen EN (1983) DMCM: a potent convulsive benzodiazepine receptor ligand. Eur J Pharmacol 94:117–124
Riedel G, Micheau J, Lam AG, Roloff E, Martin SJ, Bridge H, Hoz L, Poeschel B, McCulloch J, Morris RG (1999) Reversible neural inactivation reveals hippocampal participation in several memory processes. Nat Neurosci 2:898–905
Rudolph U, Möhler H (2004) Analysis of GABAA receptor function and dissection of the pharmacology of benzodiazepines and general anesthetics through mouse genetics. Annu Rev Pharmacol Toxicol 44:475–498
Rudolph U, Crestani F, Benke D, Brünig I, Benson JA, Fritschy J-M, Martin JR, Bluethmann H, Möhler H (1999) Benzodiazepine actions mediated by specific γ-aminobutyric acidA receptor subtypes. Nature 401:796–800
Sarter M, Stephens DN (1988) β-carbolines as tools in memory research: animal data and speculations. Psychopharmacol Ser 6:230–245
Schafe GE, LeDoux JE (2000) Memory consolidation of auditory Pavlovian fear conditioning requires protein synthesis and protein kinase A in the amygdala. J Neurosci 20:RC96, 1–5
Sieghart W (1995) Structure and pharmacology of γ-aminobutyric acidA receptor subtypes. Pharmacol Rev 47:181–234
Sieghart W, Sperk G (2002) Subunit composition, distribution and function of GABAA receptor subtypes. Curr Top Med Chem 2:795–816
Simon J, Wakimoto H, Fujita N, Lalande M, Barnard EA (2004) Analysis of the set of GABAA receptor genes in the human genome. J Biol Chem 279:41422–41435
Squire LR, Alvarez P (1995) Retrograde amnesia and memory consolidation: a neurobiological perspective. Curr Opin Neurobiol 5:169–177
Stephens DN, Schneider HH, Kehr W, Jensen LH, Petersen E, Honore T (1987) Modulation of anxiety by beta-carbolines and other benzodiazepine receptor ligands: relationship of pharmacological to biochemical measures of efficacy. Brain Res Bull 19:309–318
Stephens DN, Pistovkacova J, Worthing L, Atack JR, Dawson GR (2005) Role of GABAA α5-containing receptors in ethanol reward in the mouse: the effects of targeted gene deletion and a selective inverse agonist. Eur J Pharmacol 526:240–250
Sternfeld F, Carling RW, Jelley RA, Ladduwahetty T, Merchant KJ, Moore KW, Reeve AJ, Street LJ, O’Connor D, Sohal B, Atack JR, Cook S, Seabrook GR, Wafford KA, Tattersall FD, Collinson N, Dawson GR, Castro JL, MacLeod AM (2004) Selective, orally active γ-aminobutyric acidA α5 receptor inverse agonists as cognition enhancers. J Med Chem 47:2176–2179
Street LJ, Sternfeld F, Jelley RA, Reeve AJ, Carling RW, Moore KW, McKernan RM, Sohal B, Cook S, Pike A, Dawson GR, Bromidge FA, Wafford KA, Seabrook GR, Thompson SA, Marshall G, Pillai GV, Castro JL, Atack JR, MacLeod AM (2004) Synthesis and biological evaluation of 3-heterocyclyl-7,8,9,10-tetrahydro-(7,10-ethano)-1,2,4-triazolo[3,4-a]phthalazines and analogues as subtype-selective inverse agonists for the GABAAα5 benzodiazepine binding site. J Med Chem 47:3642–3657
Sur C, Fresu L, Howell O, McKernan RM, Atack JR (1999) Autoradiographic localization of α5 subunit-containing GABAA receptors in rat brain. Brain Res 822:265–270
Venault P, Chapouthier G, de Carvalho LP, Simiand J, Morre M, Dodd RH, Rossier J (1986) Benzodiazepine impairs and beta-carboline enhances performance in learning and memory tasks. Nature 321:864–866
Wisden W, Laurie DJ, Monyer H, Seeburg PH (1992) The distribution of 13 GABAA receptor subunit mRNAs in the rat brain. I. Telencephalon, diencephalon, mesencephalon. J Neurosci 12:1040–1062
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Collinson, N., Atack, J.R., Laughton, P. et al. An inverse agonist selective for α5 subunit-containing GABAA receptors improves encoding and recall but not consolidation in the Morris water maze. Psychopharmacology 188, 619–628 (2006). https://doi.org/10.1007/s00213-006-0361-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0361-z