Abstract
Rationale
Anticipatory nausea (AN) experienced by chemotherapy patients is resistant to current anti-nausea treatments. In this study, the effect of manipulation of the endocannabinoid (EC) system on a rat model of nausea (conditioned gaping) was determined.
Objective
The potential of cannabidiol (CBD) and the fatty acid amide hydrolase (FAAH) inhibitor, URB597 (URB) to reduce conditioned gaping in rats were evaluated.
Materials and methods
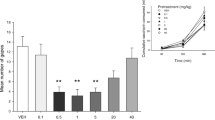
In each experiment, rats received four conditioning trials in which they were injected with lithium chloride immediately before placement in a distinctive odor-laced context. During testing, in experiment 1, rats were injected with vehicle (VEH), 1, 5 or 10 mg/kg CBD 30 min before placement in the context previously paired with nausea and in experiment 2, rats were injected with VEH, 0.1 or 0.3 mg/kg URB 2 h before placement in the context. Additional groups evaluated the ability of the CB1 antagonist/inverse agonist, SR141716A, to reverse the suppressive effects of URB. Experiment 3 measured the potential of URB to interfere with the establishment of conditioned gaping.
Results
When administered before testing, CBD (1 and 5, but not 10 mg/kg) and URB (0.3, but not 0.1 mg/kg) suppressed conditioned gaping. The effect of URB was reversed by pre-treatment with the CB1 antagonist/inverse agonist, SR141716A. When administered before conditioning, URB also interfered with the establishment of conditioned gaping.
Conclusions
Manipulations of the EC system may have therapeutic potential in the treatment of AN.



Similar content being viewed by others
References
Aapro M (2005) Optimising antiemetic therapy: what are the problems and how can they be overcome? Curr Med Res Opin 21:885–897
Cross-Mellor SK, Ossenkopp K-P, Piomelli D, Parker L (2007) Effects of the FAAH inhibitor, URB597, and anandamide on lithium-induced taste reactivity responses: a measure of nausea in the rat. Psychopharmacology 190:135–143
Darmani NA (2001) Delta-9-tetrahydrocannabinol and synthetic cannabinoids prevent emesis produced by the cannabinoid CB1 receptor antagonist/inverse agonist SR 141716A. Neuropsychopharmacology 24:198–203
Deutsch DG, Chin SA (1993) Enzymatic synthesis and degradation of anandamide, a cannabinoid receptor agonist. Biochem Pharmacol 46:791–796
Devane WA, Hanus L, Bruer A, Pertwee RG, Stevenson LA, Griffin G, Gibson D, Mandelbaum A, Etinger A, Mechoulam R (1992) Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258:1946–1949
Fegley D, Gaetani S, Duranti A, Tontini A, Mor M, Tarzia G, Piomelli D (2005) Characterization of the fatty acid amide hydrolase inhibitor cyclohexyl carbamic acid 3′-carbamoyl-biphenyl-3-yl ester (URB597): effects on anandamide and oleoylethanolamide deactivation. J Pharmacol Exp Ther 313:352–358
Foubert J, Vaessen G (2005) Nausea: the neglected symptom? Eur J Oncol Nurs 9:21–32
Hampson AJ, Grimaldi M, Axelrod J, Wink D (1998) Cannabidiol and delta-9-tetrahydorcannabinol are neuroprotective antioxciants. Proc Natl Acad Sci USA 95:8268–8273
Jordan K, Kasper C, Schmoll H-J (2005) Chemotherapy-induced nausea and vomiting: current and new standards in the antiemetic prophylaxis and treatment. Eur J Cancer 41:199–205
Kathuria S, Gaetani S, Fegley D, Valino F, Duranti A, Tontini A, Mor M, Tarzia G, La Rana GL, Calignano A, Guistino A, Tattoli M, Palmery M, Cuomo V, Piomelli D (2003) Modulation of anxiety through blockade of anandamide hydrolysis. Nat Med 9:76–81
Kwiatkowska M, Parker LA (2005) Ondansetron and delta-9-tetrahydrocannabinol interfere with the establishment of lithium-induced conditioned taste avoidance in the house musk shrew (Suncus murinus) Behav Neurosci 119:974–982
Kwiatkowska M, Parker LA, Burton P, Mechoulam R (2004) A comparative analysis of the potential of cannabinoids and ondansetron to suppress cisplatin-induced emesis in the Suncus murinus (house musk shrew). Psychopharmacology 174:254–259
Limebeer CL, Parker LA (1999) Delta-9-tetrahydrocannabinol interferes with the establishment and the expression of conditioned rejection reactions produced by cyclophosphamide: a rat model of nausea. Neuroreport 10:3769–3772
Limebeer CL, Parker LA (2000) The antiemetic drug ondansetron interferes with lithium-induced conditioned rejection reactions, but not lithium-induced taste avoidance in rats. J Exp Psychol Anim Behav Processes 26:371–384
Limebeer CL, Hall G, Parker LA (2006) Exposure to a lithium-paired context elicits gaping in rats: a model of anticipatory nausea. Physiol Behav 88:398–403
Limebeer CL, Krohn JP, Cross-Mellor S, Litt, DE, Ossenkopp K-P, Parker LA (2007) Exposure to a context previously associated with toxin (LiCl)-or motion-induced sickness elicits conditioned gaping in rats: evidence in support of a model of anticipatory nausea. Behav Brain Res, in press
Malfait AM, Gallily R, Sumariwalla PF, Malik AS, Andreakos E, Mechoulam R, Feldmann M (2000) The nonpsychoactive cannabis constituent cannabidiol is an oral anti-arthritic therapeutic in murine collagen-induced arthritis. Proc Natl Acad Sci USA 97:9561–9566
Morrow GR, Roscoe JA, Hynes HE, Flynn PJ, Pierce HI, Burish T (1998) Progress in reducing anticipatory nausea and vomiting: a study of community practice. Support Care Cancer 6:46–50
Parker LA (2003) Taste avoidance and taste aversion: evidence for two different processes. Learn Behav 31:165–172
Parker LA, Limebeer CL (2006) Conditioned gaping in rats: a selective measure of nausea. Auton Neurosci 129:36–41
Parker LA, Mechoulam R (2003) Cannabinoid agonists and antagonist modulate lithium-induced conditioned gaping in rats. Integr Physiol Behav Sci 38:133–145
Parker LA, Mechoulam R, Schlievert C (2002) Cannabidiol, a non-psychoactive component of cannabis and its synthetic dimethylheptyl homolog suppress nausea in an experimental model with rats. Neuroreport 13:567–570
Parker LA, Mechoulam R, Schlievert C, Abbott L, Fudge ML, Burton P (2003) Effects of cannabinoids on lithium-induced conditioned rejection reactions in a rat model of nausea. Psychopharmacology 166:156–162
Parker LA, Burton P, Sorge RE, Yakiwchuk C, Mechoulam R (2004a) Effect of low doses of Δ9-tetrahydrocannabinol and cannabidiol on the extinction of cocaine-induced and amphetamine-induced conditioned place preference learning in rats. Psychopharmacology 175:360–366
Parker LA, Kwiatkowska M, Burton P, Mechoulam R (2004b) Effect of cannabinoids on lithium-induced vomiting in the Suncus murinus (house musk shrew). Psychopharmacology 171:156–161
Parker LA, Kwiatkowska M, Mechoulam R (2006) Delta-9-Tetrahydrocannabinol and cannabidiol, but not ondansetron, interfere with conditioned retching reactions elicited by a lithium-paired context in Suncus murinus: an animal model of anticipatory nausea and vomiting. Physiol Behav 87:66–71
Piomelli D (2003) The molecular logic of endocannabinoid signalling. Nat Rev Neurosci 4:873–884
Travers JB, Norgren R (1986) Electromyographic analysis of the ingestion and rejection of sapid stimuli in the rat. Behav Neurosci 100:544–555
Van Sickle MD, Oland LD, Ho W, Hillard CJ, Mackie K, Davison JS, Sharkey KA (2001) Cannabinoids inhibit emesis through CB1 receptors in the brainstem of the ferret. Gastroenterology 121:767–774
Varvel SA, Wise LE, Niyuhire F, Cravatt BF, Lichtman AH (2007) Inhibition of fatty-acid amide hydrolase accelerates acquisition and extinction rates in a spatial memory task. Neuropsychopharmacology 32:1032–1041
Acknowledgments
This research was supported by a grant from the Natural Sciences and Engineering Research Council of Canada to LAP, the Israel Science Foundation and NIH to RM, NIH to DP and by a grant from Wilfrid Laurier University’s Student Technology Endowment Program and an Ontario Graduate Scholarship to EMR.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s00213-011-2332-2.
Rights and permissions
About this article
Cite this article
Rock, E.M., Limebeer, C.L., Mechoulam, R. et al. The effect of cannabidiol and URB597 on conditioned gaping (a model of nausea) elicited by a lithium-paired context in the rat. Psychopharmacology 196, 389–395 (2008). https://doi.org/10.1007/s00213-007-0970-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-007-0970-1