Abstract
Rationale
Monoamine releasers with prominent dopaminergic actions, e.g., d-methamphetamine (d-MA), significantly reduce cocaine use and craving in clinical and preclinical laboratory studies. However, D-MA and related drugs also display high abuse potential, which limits their acceptability as agonist replacement medications for the management of Cocaine Use Disorder.
Objectives
The l-isomer of methamphetamine (l-MA), unlike d-MA, has preferential noradrenergic actions and is used medicinally with low, if any, abuse liability. The present study was conducted to determine whether l-MA could serve as an agonist replacement medication by both mimicking interoceptive effects of cocaine and decreasing intravenous (IV) cocaine self-administration.
Methods
Separate groups (N = 4–5) of rhesus monkeys were studied to determine whether l-MA could (1) substitute for cocaine in subjects that discriminated intramuscular (IM) cocaine (0.4 mg/kg) from saline and (2) decrease IV cocaine self-administration under a second-order FR2(VR16:S) schedule of reinforcement.
Results
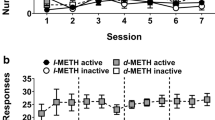
l-MA, like d-MA but with approximately 5-fold lesser potency, substituted for cocaine in drug discrimination experiments in a dose-dependent manner. In IV self-administration studies, 5–10-day treatments with continuously infused l-MA (0.032–0.32 mg/kg/h, IV) dose-dependently decreased cocaine-maintained responding; the highest dosage reduced cocaine intake to levels of saline self-administration without appreciable effects on food-maintained responding.
Conclusions
These results indicate that l-MA both shares discriminative stimulus effects with cocaine and reduces cocaine self-administration in a behaviorally selective manner. l-MA and other compounds with a similar pharmacological profile deserve further evaluation for the management of Cocaine Use Disorder.



Similar content being viewed by others
References
Banks ML, Bauer CT, Blough BE, Rothman RB, Partilla JS, Baumann MH et al (2014) Abuse-related effects of dual dopamine/serotonin releasers with varying potency to release norepinephrine in male rats and rhesus monkeys. Exp Clin Psychopharmacol 22:274–284. doi:10.1037/a0036595
Baumann MH, Clark RD, Woolverton WL, Wee S, Blough BE, Rothman RB (2011a) In vivo effects of amphetamine analogs reveal evidence for serotonergic inhibition of mesolimbic dopamine transmission in the rat. J Pharmacol Exp Ther 337:218–225. doi:10.1124/jpet.110.176271
Baumann MH, Williams Z, Zolkowska D, Rothman RB (2011b) Serotonin (5-HT) precursor loading with 5-hydroxy-l-tryptophan (5-HTP) reduces locomotor activation produced by (+)-amphetamine in the rat. Drug Alcohol Depend 114:147–152. doi:10.1016/j.drugalcdep.2010.09.015
Cunningham JK, Maxwell JC, Campollo O, Liu LM, Lattyak WJ, Callaghan RC (2013) Mexico’s precursor chemical controls: emergence of less potent types of methamphetamine in the United States. Drug Alcohol Depend 129:125–136. doi:10.1016/j.drugalcdep.2012.10.001
Czoty PW, Makriyannis A, Bergman J (2004a) Methamphetamine discrimination and in vivo microdialysis in squirrel monkeys. Psychopharmacology (Berl) 175:170–178. doi:10.1007/s00213-004-1798-6
Czoty PW, Ramanathan CR, Mutschler NH, Makriyannis A, Bergman J (2004b) Drug discrimination in methamphetamine-trained monkeys: effects of monoamine transporter inhibitors. J Pharmacol Exp Ther 311:720–727. doi:10.1124/jpet.104.071035
Czoty PW, Gould RW, Martelle JL, Nader MA (2010a) Prolonged attenuation of the reinforcing strength of cocaine by chronic d-amphetamine in rhesus monkeys. Neuropsychopharmacology 36:539–547. doi:10.1038/npp.2010.185
Czoty PW, Martelle JL, Nader MA (2010b) Effects of chronic d-amphetamine administration on the reinforcing strength of cocaine in rhesus monkeys. Psychopharmacology (Berl) 209:375–382. doi:10.1007/s00213-010-1807-x
Desai RI, Bergman J (2010) Drug discrimination in methamphetamine-trained rats: effects of cholinergic nicotinic compounds. J Pharmacol Exp Ther 335:807–816. doi:10.1124/jpet.110.173773
Dufka F, Galloway G, Baggott M, Mendelson J (2009) The effects of inhaled L-methamphetamine on athletic performance while riding a stationary bike: a randomised placebo-controlled trial. Br J Sports Med 43:832–835. doi:10.1136/bjsm.2008.048348
Fowler JS, Kroll C, Ferrieri R, Alexoff D, Logan J, Dewey SL et al (2007) PET studies of d-methamphetamine pharmacokinetics in primates: comparison with l-methamphetamine and (-)-cocaine. J Nucl Med 48:1724–1732. doi:10.2967/jnumed.107.040279
Grabowski J, Shearer J, Merrill J, Negus SS (2004) Agonist-like, replacement pharmacotherapy for stimulant abuse and dependence. Addict Behav 29:1439–1464. doi:10.1016/j.addbeh.2004.06.018
Herin DV, Rush CR, Grabowski J (2010) Agonist-like pharmacotherapy for stimulant dependence: preclinical, human laboratory, and clinical studies. Ann N Y Acad Sci 1187:76–100. doi:10.1111/j.1749-6632.2009.05145.x
ILAR-NRC (2011) Guide for the care and use of laboratory animals, 8th edition (Institute of Laboratory Animal Resources, Commission on Life Sciences, National Research Council). The National Academies press 125
Kirkpatrick MG, Gunderson EW, Perez AY, Haney M, Foltin RW, Hart CL (2012) A direct comparison of the behavioral and physiological effects of methamphetamine and 3,4-methylenedioxymethamphetamine (MDMA) in humans. Psychopharmacology (Berl) 219:109–122. doi:10.1007/s00213-011-2383-4
Kohut SJ, Riley AL (2013) Conditioned taste aversions. In: Stolerman (ed) Encyclopedia of Psychopharmacology
Kohut SJ, Fivel PA, Blough BE, Rothman RB, Mello NK (2013a) Effects of methcathinone and 3-Cl-methcathinone (PAL-434) in cocaine discrimination or self-administration in rhesus monkeys. Int J Neuropsychopharmacol 16:1985–1998. doi:10.1017/S146114571300059X
Kohut SJ, Fivel PA, Mello NK (2013b) Differential effects of acute and chronic treatment with the α2-adrenergic agonist, lofexidine, on cocaine self-administration in rhesus monkeys. Drug Alcohol Depend 133:593–599. doi:10.1016/j.drugalcdep.2013.07.032
Kuczenski R, Segal DS, Cho AK, Melega W (1995) Hippocampus norepinephrine, caudate dopamine and serotonin, and behavioral responses to the stereoisomers of amphetamine and methamphetamine. J Neurosci 15:1308–1317
Li L, Everhart T, Jacob P III, Jones R, Mendelson J (2010) Stereoselectivity in the human metabolism of methamphetamine. Br J Clin Pharmacol 69:187–192. doi:10.1111/j.1365-2125.2009.03576.x
McCabe SE, Teter CJ, Boyd CJ (2006) Medical use, illicit use and diversion of prescription stimulant medication. J Psychoactive Drugs 38:43–56
Melega WP, Cho AK, Schmitz D, Kuczenski R, Segal DS (1999) l-methamphetamine pharmacokinetics and pharmacodynamics for assessment of in vivo deprenyl-derived l-methamphetamine. J Pharmacol Exp Ther 288:752–758
Mello NK, Fivel PA, Kohut SJ (2013) Effects of chronic buspirone treatment on nicotine and concurrent nicotine + cocaine self-administration. Neuropsychopharmacology 38:1264–1275
Mendelson J, Uemura N, Harris D, Nath RP, Fernandez E, Jacob P III et al (2006) Human pharmacology of the methamphetamine stereoisomers. Clin Pharmacol Ther 80:403–420. doi:10.1016/j.clpt.2006.06.013
Mendelson JE, McGlothlin D, Harris DS, Foster E, Everhart T, Jacob P III et al (2008) The clinical pharmacology of intranasal l-methamphetamine. PLoS One 8:4. doi:10.1186/1472-6904-8-4
Mooney ME, Herin DV, Schmitz JM, Moukaddam N, Green CE, Grabowski J (2009) Effects of oral methamphetamine on cocaine use: a randomized, placebo-controlled trial. Drug Alcohol Depend 101:34–41
Negus SS, Henningfield J (2014) Agonist medications for the treatment of Cocaine Use Disorder. Neuropsychopharmacology 40:1815–1825. doi:10.1038/npp.2014.322
Negus SS, Mello NK (2003) Effects of chronic d-amphetamine treatment on cocaine-and food-maintained responding under a second-order schedule in rhesus monkeys. Drug Alcohol Depend 70:39–52. doi:10.1016/S0376-8716(02)00339-3
Negus SS, Mello NK, Blough BE, Baumann MH, Rothman RB (2007) Monoamine releasers with varying selectivity for dopamine/norepinephrine versus serotonin release as candidate “agonist” medications for cocaine dependence: studies in assays of cocaine discrimination and cocaine self-administration in rhesus monkeys. J Pharmacol Exp Ther 320:627–636. doi:10.1124/jpet.106.107383
Negus SS, Baumann MH, Rothman RB, Mello NK, Blough BE (2009) Selective suppression of cocaine- versus food-maintained responding by monoamine releasers in rhesus monkeys: benzylpiperazine, (+)phenmetrazine, and 4-benzylpiperidine. J Pharmacol Exp Ther 329:272–281. doi:10.1124/jpet.108.143701
Rabiner DL, Anastopoulos AD, Costello EJ, Hoyle RH, McCabe SE, Swartzwelder HS (2009) The misuse and diversion of prescribed ADHD medications by college students. J Atten Disord 13:144–153. doi:10.1177/1087054708320414
Riley AL (2011) The paradox of drug taking: the role of the aversive effects of drugs. Physiol Behav 103(1):69–78
Rothman RB, Baumann MH (2003) Monoamine transporters and psychostimulant drugs. Eur J Pharmacol 479:23–40. doi:10.1016/j.ejphar.2003.08.054
Rothman RB, Baumann MH (2006) Balance between dopamine and serotonin release modulates behavioral effects of amphetamine‐type drugs. Ann N Y Acad Sci 1074:245–260
Rothman RB, Baumann MH, Dersch CM, Romero DV, Rice KC, Carroll FI, Partilla JS (2001) Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin. Synapse 39:32–41. doi:10.1002/1098-2396(20010101)39:1<32::AID-SYN5>3.0.CO;2-3
SAMHSA (2014) Substance abuse and mental health services administration. Results from the 2013 national survey on drug use and health: summary of national findings, NSDUH series H-48, HHS publication No. (SMA) 14-4863. Substance abuse and mental health services administration, Rockville
Spealman RD (1995) Noradrenergic involvement in the discriminative stimulus effects of cocaine in squirrel monkeys. J Pharmacol Exp Ther 275:53–62
Stoops WW, Rush CR (2013) Agonist replacement for stimulant dependence: a review of clinical research. Curr Pharm Des 19:7026–7035
Upadhyaya HP, Rose K, Wang W, O’Rourke K, Sullivan B, Deas D, Brady KT (2005) Attention-deficit/hyperactivity disorder, medication treatment, and substance use patterns among adolescents and young adults. J Child Adolesc Psychopharmacol 15:799–809. doi:10.1089/cap.2005.15.799
Winger GD, Yasar S, Negus SS, Goldberg SR (1994) Intravenous self-administration studies with l-deprenyl (selegiline) in monkeys. Clin Pharmacol Ther 56:774–780
Acknowledgments
This work was supported by grants R01-DA002519 and K01-DA039306 from the National Institute on Drug Abuse, National Institute of Health. We thank Claire Barkin, Nathanial Osiris, and Olga Smirnova for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Kohut, S.J., Bergman, J. & Blough, B.E. Effects of l-methamphetamine treatment on cocaine- and food-maintained behavior in rhesus monkeys. Psychopharmacology 233, 1067–1075 (2016). https://doi.org/10.1007/s00213-015-4186-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-015-4186-5