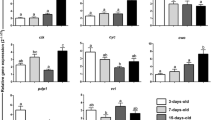
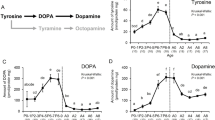
Abstract
Octopamine (OA) and juvenile hormone (JH) are implicated in the regulation of age-based division of labor in the honeybee, Apis mellifera. We tested the hypothesis that these two neuroendocrine signals influence task-associated plasticity in circadian and diurnal rhythms, and in brain expression of the clock gene period (per). Treatment with OA, OA antagonist (epinastine), or both, did not affect the age at onset of circadian rhythmicity or the free running period in constant darkness (DD). Young bees orally treated with OA in light–dark (LD) illumination regime for 6 days followed by DD showed reduced alpha (the period between the daily onset and offset of activity) during the first 4 days in LD and the first 4 days in DD. Oral treatment with OA, epinastine, or both, but not manipulations of JH levels, caused increased average daily levels and aberrant patterns of brain per mRNA oscillation in young bees. These results suggest that OA and JH do not influence the development or function of the central pacemaker but rather that OA influences the brain expression of a clock gene and characteristics of locomotor behavior that are not thought to be under direct control of the circadian pacemaker.





Similar content being viewed by others
Abbreviations
- ANOVA:
-
Analysis of variance
- 5-HT:
-
Serotonin
- CA:
-
Corpora allata
- CT:
-
Circadian time
- DA:
-
Dopamine
- DD:
-
Constant darkness
- DHBA:
-
Dihydroxybenzylamine
- EF-1α:
-
Elongation factor-1α
- Epi:
-
Epinastine
- HPLC:
-
High-performance liquid chromatography
- FRP:
-
Free running period
- JH:
-
Juvenile hormone
- LD:
-
Light–dark
- Met:
-
Methoprene
- OA:
-
Octopamine
- PCR:
-
Polymerase chain reaction
- Per:
-
Period
- RT:
-
Reverse transcription
- SCN:
-
Suprachiasmatic nucleus
- SS:
-
Sugar syrup
- ZT:
-
Zeitgeber time
References
Barron AB, Robinson GE (2005) Selective modulation of task performance by octopamine in honey bee (Apis mellifera) division of labour. J Comp Physiol A 191:659–668
Barron AB, Schulz DJ, Robinson GE (2002) Octopamine modulates responsiveness to foraging-related stimuli in honey bees (Apis mellifera). J Comp Physiol A 188:603–610
Battelle BA (2002) Circadian efferent input to Limulus eyes: anatomy, circuitry, and impact. Microsc Res Techniq 58:345–355
Bicker G (1999) Biogenic amines in the brain of the honeybee: cellular distribution, development, and behavioral functions. Microsc Res Techniq 44:166–178
Bicker G, Menzel R (1989) Chemical codes for the control of behaviour in arthropods. Nature, 337:33–39
Bloch G, Robinson GE (2001) Reversal of honeybee behavioural rhythms. Nature 410:1048
Bloch G, Toma DP, Robinson GE (2001) Behavioral rhythmicity, age, division of labor and period expression in the honey bee brain. J Biol Rhythms 16:444–456
Bloch G, Wheeler DE, Robinson GE (2002a) Endocrine influences on the organization of insect societies. In: Pfaff D, rnold AP, Etgen AM, Fahrbach SE, Rubin RT (eds) Hormones, Brain and Behavior, vol. III, Non-mammalian hormone-behavior systems. Academic, San Diego, pp 195–235
Bloch G, Sullivan JP, Robinson GE (2002b) Juvenile hormone and circadian locomotor activity in the honey bee Apis mellifera. J Insect Physiol 48:1123–1131
Bloch G, Solomon SM, Robinson GE, Fahrbach SE (2003) Patterns of PERIOD and pigment-dispersing hormone immunoreactivity in the brain of the European honeybee (Apis mellifera): age- and time-related plasticity. J Comp Neurol 464:269–284
Bloch G, Rubinstein CD, Robinson GE (2004) Period expression in the honey bee brain is developmentally regulated and not affected by light, flight experience, or colony type. Insect Biochem Mol Biol 34:879–891
Bloch G, Shemesh Y, Robinson GE (2006) Seasonal and task-related variation in free running activity rhythms in honey bees (Apis mellifera). Insect Soc 53:115–118
Burrell BD, Smith BH (1995) Modulation of the honey bee (Apis mellifera) sting response by octopamine. J Insect Physiol 41:671–680
Chyb S, Hevers W, Forte M, Wolfgang WJ, Selinger Z, Hardie RC (1999) Modulation of the light response by cAMP in Drosophila photoreceptors. J Neurosci 19:8799–8807
Cymborowski B (1998) Serotonin modulates a photic response in circadian locomotor rhythmicity of adults of the blow fly, Calliphora vicina. Physiol Entomol 23:25–32
Degen J, Gewecke M, Roeder T (2000) Octopamine receptors in the honey bee and locust nervous system: pharmacological similarities between homologous receptors of distantly related species. Br J Pharmacol 130:587–594
Drijfhout WJ, van der Linde AG, Kooi SE, Grol CJ, Westerink BHC (1996) Norepinephrine release in the rat pineal gland: the input from the biological clock measured by in vivo microdialysis. J Neurochem 66:748–755
Dunlap JC (1999) Molecular bases for circadian clocks. Cell 96:271–290
Erber J, Kloppenburg P, Scheidler A (1993) Neuromodulation by serotonin and octopamine in the honeybee—behaviour, neuroanatomy and electrophysiology. Experientia 49:1073–1083
Franken P, Dudley CA, Estill SJ, Barakat M, Thomason R, O’Hara BF, McKnight SL (2006) NPAS2 as a transcriptional regulator of non-rapid eye movement sleep: genotype and sex interactions. Proc Nat Acad Sci USA 103:7118–7123
Fussnecker BL, Smith BH, Mustard JA (2006) Octopamine and tyramine influence the behavioral profile of locomotor activity in the honey bee (Apis mellifera). J Insect Physiol DOI 10.1016/j.jinsphys.2006.07.008 (in press)
Gingras JL, Lawson EE, McNamara MC (1996) Developmental characteristics in the daily rhythm of norepinephrine concentration within rabbit brainstem regions. Reprod Fert Dev 8:189–194
Giray T, Huang ZY, Guzman-Novoa E, Robinson GE (1999) Physiological correlates of genetic variation for rate of behavioral development in the honeybee, Apis mellifera. Behav Ecol Sociobiol 47:17–28
Goel N, Governale MM, Jechura TJ, Lee TM (2000) Effects of intergeniculate leaflet lesions on circadian rhythms in Octodon degus. Brain Res 877:306–313
Gracioli LF, de Moraes RLMS (2002) Juvenile hormone promotes changes in the expression of hypopharyngeal gland proteins of worker Apis mellifera (Hymenoptera:Apidae). Sociobiology 40:443–448
Hall JC (1998) Genetics of biological rhythms in Drosophila. Adv Genet 38:135–184
Huang ZY, Robinson GE, Tobe SS, Yagi KJ, Strambi C, Strambi A, Stay B (1991) Hormonal regulation of behavioural development in the honey bee is based on changes in the rate of juvenile hormone biosynthesis. J Insect Physiol 37:733–742
Kaiser W, Steiner-Kaiser J (1983) Neuronal correlates of sleep, wakefulness and arousal in a diurnal insect. Nature 301:707–709
Kaiser W (1988) Busy bees need rest, too: Behavioral and electromyographical sleep signs in honeybees. J Comp Physiol A 163:565–584
Khadilkar RV, Mytinger JR, Thomason LE, Runyon SL, Washicosky KJ, Jinks RN (2002) Central regulation of photosensitive membrane turnover in the lateral eye of Limulus. I. Octopamine primes the retina for daily transient rhabdom shedding. Vis Neurosci 19:283–297
Klarsfeld A, Leloup JC, Rouyer F (2003) Circadian rhythms of locomotor activity in Drosophila. Behav Proc 64:161–175
Kloppenburg P, Erber J (1995) The modulatory effects of serotonin and octopamine in the visual system of the honey bee (Apis mellifera l) .2. Electrophysiological analysis of motion-sensitive neurons in the lobula. J Comp Physiol A 176:119–129
Kubo T, Sasaki M, Nakamura J, Sasagawa H, Ohashi K, Takeuchi H, Natori S (1996) Change in the expression of hypopharyngeal-gland proteins of the worker honeybees (Apis mellifera L.) with age and/or role. J Biochem Tokyo 119:291–295
Lamont EW, Robinson B, Stewart J, Amir S (2005) The central and basolateral nuclei of the amygdala exhibit opposite diurnal rhythms of expression of the clock protein Period2. Proc Nat Acad Sci USA 102:4180–4184
Linn CE, Roelofs WL (1986) Modulatory effects of octopamine and serotonin on male sensitivity and periodicity of response to sex pheromones in the cabbage looper moth, Trichoplusia ni. Arch Insect Biochem Physiol 3:161–171
Linn CE, Roelofs WL (1992) Role of photoperiod cues in regulating the modulatory action of octopamine on pheromone-response thresholds in the cabbage-looper moth. Arc Insect Biochem Physiol 20:285–302
Linn CE, Campbell MG, Poole KR, Wu WQ, Roelofs WL (1996) Effects of photoperiod on the circadian timing of pheromone response in male Trichoplusia ni: relationship to the modulatory action of octopamine. J Insect Physiol 42:881–891
Lohse MJ (1993) Molecular mechanisms of membrane receptor desensitization. Biochim Biophys Acta 1179:171–188
Maqueira B, Chatwin H, Evans PD (2005) Identification and characterization of a novel family of Drosophila beta-adrenergic-like octopamine G-protein coupled receptors. J Neurochem 94:547–560
Menzel R, Muller U (1996) Learning and memory in honeybees: from behavior to neural substrates. Annu Rev Neurosci 19:379–404
Meyer-Bernstein EL, Morin LP (1996) Differential serotonergic innervation of the suprachiasmatic nucleus and the intergeniculate leaflet and its role in circadian rhythm modulation. J Neurosci 16:2097–2111
Meyer-Bernstein EL, Blanchard JH, Morin LP (1997) The serotonergic projection from the median raphe nucleus to the suprachiasmatic nucleus modulates activity phase onset, but not other circadian rhythm parameters. Brain Res 25:112–120
Milde JJ, Homberg U (1984) Ocellar interneurons in the honeybee - characteristics of spiking l-neurons. J Comp Physiol A 155:151–160
Meinertzhagen IA, Pyza E (1999) Neurotransmitter regulation of circadian structural changes in the fly’s visual system. Microsc Res Techniq 45:96–105
Mizunami M (1995) Functional diversity of neural organization in insect ocellar systems. Vis Res 35:443–452
Moore D (2001) Honey bee circadian clocks: behavioral control from individual workers to whole-colony rhythms. J Insect Physiol 47:843–857
Moore D, Angel JE, Cheeseman IM, Fahrbach SE, Robinson GE (1998) Timekeeping in the honey bee colony: integration of circadian rhythms and division of labor. Behav Ecol Sociobiol 43:147–160
Morin LP (1999) Serotonin and the regulation of mammalian circadian rhythmicity. Ann Med 31:12–33
Moritz RFA, Sakofski F (1991) The role of the queen in circadian rhythems of honeybees (Apis mellifera L.). Behav Ecol Sociobiol 29:361–365
Nakamura TJ, Moriya T, Inoue S, Shimazoe T, Watanabe S, Ebihara S, Shinohara K (2005) Estrogen differentially regulates expression of Per1 and Per2 genes between central and peripheral clocks and between reproductive and nonreproductive tissues in female rats. J Neurosci Res 82:622–630
Ohashi K, Natori S, Kubo T (1997) Change in the mode of gene expression of the hypopharyngeal gland cells with an age-dependent role change of the worker honeybee Apis mellifera L. Eur J Biochem 249:797–802
Pankiw T, Page RE (2003) Effect of pheromones, hormones, and handling on sucrose response thresholds of honey bees (Apis mellifera L.). J Comp Physiol A 189:675–684
Peitsch D, Fietz A, Hertel H, Desouza J, Ventura DF, Menzel R (1992) The spectral input systems of hymenopteran insects and their receptor-based color-vision. J Comp Physiol A 170:23–40
Perrin JS, Segall LA, Harbour VL, Woodside B, Amir S (2006) The expression of the clock protein PER2 in the limbic forebrain is modulated by the estrous cycle. Proc Nat Acad Sci USA 103:5591–5596
Pophof B (2000) Octopamine modulates the sensitivity of silkmoth pheromone receptor neurons. J Comp Physiol A 186:307–313
Renner M (1955) Ein Transozeanversuch zum Zeitsinn der Honigbiene. Naturwissenchaften 42:540–541
Robinson GE (1985) Effects of a juvenile hormone analogue on honey bee foraging behaviour and alarm pheromone production. J Insect Physiol 31:277–282
Robinson GE (1987) Modulation of alarm pheromone perception in the honey bee: evidence for division of labor based on hormonally regulated response thresholds. J Comp Physiol A 160:613–619
Robinson GE, Heuser LM, Le Conte Y, Lenquette F, Hollingworth RM (1999) Neurochemicals aid bee nestmate recognition. Nature 399:534–535
Roeder T (1999) Octopamine in invertebrates. Prog Neurobiol 59:533–561
Roeder T (2005) Tyramine and octopamine: ruling behavior and metabolism. Annu Rev Entomol 50:447–477
Roeder T, Degen J, Gewecke M (1998) Epinastine, a highly specific antagonist of insect neural octopamine receptors. Eur J Pharmacol 349:171–177
Rubin R, Shemesh Y, Cohen M, Elgavish S. Robertson HM, Bloch G (2006) Molecular and phylogenetic analyses reveal mammalian-like clockwork in the honey bee (Apis mellifera) and shed new light on the molecular evolution of the circadian clock. Genome Res 16:1352–1365
Saifullah ASM, Tomioka K (2002) Serotonin sets the day state in the neurons that control coupling between the optic lobe circadian pacemakers in the cricket Gryllus bimaculatus. J Exp Biol 205:1305–1314
Sasagawa H, Sasaki M, Okada I (1989) Hormonal-control of the division of labor in adult honeybees (Apis mellifera L). 1. Effect of methoprene on corpora allata and hypopharyngeal gland, and its alpha-glucosidase activity. Appl Entomol Zool 24:66–77
Scheiner R, Pluckhahn S, Oney B, Blenau W, Erber J (2002) Behavioural pharmacology of octopamine, tyramine and dopamine in honey bees. Behav Brain Res 136:545–553
Schulz DJ, Robinson GE (1999) Biogenic amines and division of labor in honey bee colonies: behaviorally related changes in antennal lobes and age-related changes in the mushroom bodies. J Comp Physiol A 184:481–488
Schulz DJ, Robinson GE (2001) Octopamine influences division of labor in honey bee colonies, J Comp Physiol A 187:53–61
Schulz DJ, Barron AB, Robinson GE (2002a). A role for octopamine in honey bee division of labor. Brain Behav Evol 60:350–359
Schulz DJ, Sullivan JP, Robinson GE (2002b) Juvenile hormone and octopamine in the regulation of division of labor in honey bee colonies. Horm Behav 42:222–231
Schulz DJ, Elekonich MM, Robinson GE (2003) Biogenic amines in the antennal lobes and the initiation and maintenance of foraging behavior in honey bees. J Neurobiol 54:406–416
Shimizu I, Kawai Y, Taniguchi M, Aoki S (2001) Circadian rhythm and cDNA cloning of the clock gene period in the honeybee Apis cerana japonica. Zool Sci 18:779–789
Sinakevitch I, Niwa M, Strausfeld NJ (2005) Octopamine-like immunoreactivity in the honey bee and cockroach: comparable organization in the brain and subesophageal ganglion. J Comp Neurol 488:233–254
Spivak M, Masterman R, Ross R, Mesce KA (2003) Hygienic behavior in the honey bee (Apis mellifera L.) and the modulatory role of octopamine. J Neurobiol 55:341–354
Sokal RR, Rohlf FJ (1995) Biometry, 3 edn. W. H. Freeman and Company, New York
Steenhard BM, Besharse JC (2000) Phase shifting the retinal circadian clock: xPer2 mRNA induction by light and dopamine. J Neurosci 20:8572–8577
Stern M, Thompson KSJ, Zhou P, Watson DG, Midgley JM, Gewecke M, Bacon JP (1995) Octopaminergic neurons in the locust brain—morphological, biochemical and electrophysiological characterization of potential modulators of the visual-system. J Comp Physiol A 177:611–625
Stevenson PA, Dyakonova V, Rillich J, Schildberger K (2005) Octopamine and experience-dependent modulation of aggression in crickets. J Neurosci 25:1431–1441
Su YF, Harden TK, Perkins JP (1980) Catecholamine-specific desensitization of adenylate-cyclase—evidence for a multistep process. J Biol Chem 255:7410–7419
Sullivan JP, Jassim O, Fahrbach SE, Robinson GE (2000) Juvenile hormone paces behavioral development in the adult worker honey bee. Horm Behav 37:1–14
Sullivan JP, Fahrbach SE, Harrison JF, Capaldi EA, Fewell JH, Robinson GE (2003) Juvenile hormone and division of labor in honey bee colonies: effects of allatectomy on flight behavior and metabolism. J Exp Biol 206:2287–2296
Takahashi S, Yokota S, Hara R, Kobayashi T, Akiyama M, Moriya T, Shibata S (2001) Physical and inflammatory stressors elevate circadian clock gene mPer1 mRNA levels in the paraventricular nucleus of the mouse. Endocrinology 142:4910–4917
Terazono H, Mutoh T, Yamaguchi S, Kobayashi M, Akiyama M, Udo R, Ohdo S, Okamura H, Shibata S (2003) Adrenergic regulation of clock gene expression in mouse liver. Proc Nat Acad Sci USA 100:6795–6800
Toma DP, Bloch G, Moore D, Robinson GE (2000) Changes in period mRNA levels in the brain and division of labor in honey bee colonies. Proc Nat Acad Sci USA 97:6914–6919
Unoki S, Matsumoto Y, Mizunami M (2005) Participation of octopaminergic reward system and dopaminergic punishment system in insect olfactory learning revealed by pharmacological study. Eur J Neurosci 22:1409–1416
von Frisch K (1967) The dance language and orientation of bees. Harvard University Press, Cambridge
Wagener-Hulme C, Kuehn JC, Schulz DJ, Robinson GE (1999) Biogenic amines and division of labor in honey bee colonies. J Comp Physiol A 184:471–479
Williams JA, Sehgal A (2001) Molecular components of the circadian system in Drosophila. Ann Rev Physiol 63:729–755
Winer J, Jung CKS, Shackel I, Williams PM (1999) Development and validation of real-time quantitative reverse transcriptase-polymerase chain reaction for monitoring gene expression in cardiac myocytes in vitro. Anal Biochem 270:41–49
Withers GS, Fahrbach SE, Robinson GE (1995) Effects of experience and juvenile hormone on the organization of the mushroom bodies of honey bees. J Neurobiol 26:130–144
Wyatt GR, Davey KG (1996). Cellular and molecular actions of juvenile hormone. II. Roles of juvenile hormone in adult insects. Adv Insect Physiol 26:2–155
Yellman C, Tao H, He B, Hirsh J (1997) Conserved and sexually dimorphic behavioral responses to biogenic amines in decapitated Drosophila. Proc Nat Acad Sci USA 94:4131–4136
Yerushalmi S, Bodenhaimer S, Bloch G (2006) Developmentally determined attenuation in circadian rhythms links chronobiology to social organization in bees. J Exp Biol 209:1044–1051
Yuan Q, Lin F, Zheng X, Sehgal A (2005) Serotonin modulates circadian entrainment in Drosophila. Neuron 47:115–127
Acknowledgments
We thank Rafi Nir and Allan Ross for expert assistance with bees; Mira Cohen, Rachel Saf, and Sara O’Brien for technical assistance in the laboratory; Bryan A. White for gracious use of the ABI 5700 real-time PCR machine. Thanks to Gene E. Robinson for support, helpful discussions, and for reading an earlier version of this manuscript, Joseph P. Sullivan for performing the allaetectomy operations, Dave J. Schulz for performing HPLC analyses for biogenic amine levels, and two anonymous reviewers for helpful comments on a previous version of this manuscript. Financial support was provided by NIH grant GM57196 to GER, the National Institute for Psychobiology in Israel grant number 036.7207 to GB, and the Israel–US Binational Science Foundation # 2001022 to GB and GER.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Bloch, G., Meshi, A. Influences of octopamine and juvenile hormone on locomotor behavior and period gene expression in the honeybee, Apis mellifera . J Comp Physiol A 193, 181–199 (2007). https://doi.org/10.1007/s00359-006-0179-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-006-0179-5