Abstract
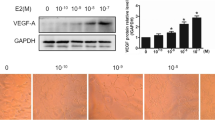
The G protein-coupled oestrogen receptor GPER1, also known as GPR30, has been implicated in oestrogen signalling, but the physiological importance of GPER1 is not fully understood. The GPER1 agonist G-1 has become an important tool to assess GPER1-mediated cellular effects. Here, we report that this substance, besides acting via GPER1, affects the microtubule network in endothelial cells. Treatment with G-1 (3 μM) for 24 h reduced DNA synthesis by about 60 % in mouse microvascular endothelial bEnd.3 cells. Treatment with 3 μM G-1 prevented outgrowth of primary endothelial cells from mouse aortic explants embedded in Matrigel. Treatment with G-1 (0.3–3 μM) for 24 h disrupted bEnd.3 cell and HUVEC microtubule structure in a concentration-dependent manner as assessed by laser-scanning confocal immunofluorescence microscopy. G-1-induced (3 μM) disruption of microtubule was observed also after acute (3 and 6 h) treatment and in the presence of the protein synthesis inhibitor cycloheximide. Disruption of microtubules by 3 μM G-1 was observed in aortic smooth muscle cells obtained from both GPER1 knockout and wild-type mice, suggesting that G-1 influences microtubules through a mechanism independent of GPER1. G-1 dose dependently (10–50 μM) stimulated microtubule assembly in vitro. On the other hand, microtubules appeared normal in the presence of 10–50 μM G-1 as determined by electron microscopy. We suggest that G-1-promoted endothelial cell anti-proliferation is due in part to alteration of microtubule organization through a mechanism independent of GPER1. This G-1-promoted mechanism may be used to block unwanted endothelial cell proliferation and angiogenesis such as that observed in, e.g. cancer.









Similar content being viewed by others
References
Green S, Walter P, Kumar V, Krust A, Bornert JM, Argos P, Chambon P (1986) Human oestrogen receptor cDNA: sequence, expression and homology to v-erb-A. Nature 320:134–139
Greene GL, Gilna P, Waterfield M, Baker A, Hort Y, Shine J (1986) Sequence and expression of human estrogen receptor complementary DNA. Science 231:1150–1154
Kuiper GG, Enmark E, Pelto-Huikko M, Nilsson S, Gustafsson JA (1996) Cloning of a novel receptor expressed in rat prostate and ovary. Proc Natl Acad Sci USA 93:5925–5930
Revankar CM, Cimino DF, Sklar LA, Arterburn JB, Prossnitz ER (2005) A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science 307:1625–1630
Thomas P, Pang Y, Filardo EJ, Dong J (2005) Identity of an estrogen membrane receptor coupled to a G protein in human breast cancer cells. Endocrinology 146:624–632
Owman C, Blay P, Nilsson C, Lolait SJ (1996) Cloning of human cDNA encoding a novel heptahelix receptor expressed in Burkitt’s lymphoma and widely distributed in brain and peripheral tissues. Biochem Biophys Res Commun 228:285–292
Nilsson S, Makela S, Treuter E, Tujague M, Thomsen J, Andersson G, Enmark E, Pettersson K, Warner M, Gustafsson JA (2001) Mechanisms of estrogen action. Physiol Rev 81:1535–1565
Isensee J, Meoli L, Zazzu V, Nabzdyk C, Witt H, Soewarto D, Effertz K, Fuchs H, Gailus-Durner V, Busch D, Adler T, de Angelis MH, Irgang M, Otto C, Noppinger PR (2009) Expression pattern of Gpr30 in LacZ reporter mice. Endocrinology 150:1722–1730
Martensson UE, Salehi SA, Windahl S, Gomez MF, Sward K, Daszkiewicz-Nilsson J, Wendt A, Andersson N, Hellstrand P, Grande PO, Owman C, Rosen CJ, Adamo ML, Lundquist I, Rorsman P, Nilsson BO, Ohlsson C, Olde B, Leeb-Lundberg LM (2009) Deletion of the G protein-coupled receptor 30 impairs glucose tolerance, reduces bone growth, increases blood pressure, and eliminates estradiol-stimulated insulin release in female mice. Endocrinology 150:687–698
Otto C, Fuchs I, Kauselmann G, Kern H, Zevnik B, Andreasen P, Schwarz G, Altmann H, Klewer M, Schoor M, Vonk R, Fritzemeier KH (2009) GPR30 does not mediate estrogenic responses in reproductive organs in mice. Biol Reprod 80:34–41
Olde B, Leeb-Lundberg LM (2009) GPR30/GPER1: searching for a role in estrogen physiology. Trends Endocrinol Metab 20:409–416
Chen Z, Yuhanna IS, Galcheva-Gargova Z, Karas RH, Mendelsohn ME, Shaul PW (1999) Estrogen receptor alpha mediates the nongenomic activation of endothelial nitric oxide synthase by estrogen. J Clin Invest 103:401–406
Darblade B, Pendaries C, Krust A, Dupont S, Fouque MJ, Rami J, Chambon P, Bayard F, Arnal JF (2002) Estradiol alters nitric oxide production in the mouse aorta through the alpha-, but not beta-, estrogen receptor. Circ Res 90:413–419
Pare G, Krust A, Karas RH, Dupont S, Aronovitz M, Chambon P, Mendelsohn ME (2002) Estrogen receptor-alpha mediates the protective effects of estrogen against vascular injury. Circ Res 90:1087–1092
Pendaries C, Darblade B, Rochaix P, Krust A, Chambon P, Korach KS, Bayard F, Arnal JF (2002) The AF-1 activation-function of ERalpha may be dispensable to mediate the effect of estradiol on endothelial NO production in mice. Proc Natl Acad Sci USA 99:2205–2210
Simoncini T, Hafezi-Moghadam A, Brazil DP, Ley K, Chin WW, Liao JK (2000) Interaction of oestrogen receptor with the regulatory subunit of phosphatidylinositol-3-OH kinase. Nature 407:538–541
Nilsson BO (2007) Modulation of the inflammatory response by estrogens with focus on the endothelium and its interactions with leukocytes. Inflamm Res 56:269–273
Toutain CE, Filipe C, Billon A, Fontaine C, Brouchet L, Guery JC, Gourdy P, Arnal JF, Lenfant F (2009) Estrogen receptor alpha expression in both endothelium and hematopoietic cells is required for the accelerative effect of estradiol on reendothelialization. Arterioscler Thromb Vasc Biol 29:1543–1550
Haas E, Bhattacharya I, Brailoiu E, Damjanovic M, Brailoiu GC, Gao X, Mueller-Guerre L, Marjon NA, Gut A, Minotti R, Meyer MR, Amann K, Ammann E, Perez-Dominguez A, Genoni M, Clegg DJ, Dun NJ, Resta TC, Prossnitz ER, Barton M (2009) Regulatory role of G protein-coupled estrogen receptor for vascular function and obesity. Circ Res 104:288–291
Lindsey SH, Cohen JA, Brosnihan KB, Gallagher PE, Chappell MC (2009) Chronic treatment with the Gpr30 agonist G-1 decreases blood pressure in ovariectomized Mren2.Lewis rats. Endocrinology 150:3753–3758
Bologa CG, Revankar CM, Young SM, Edwards BS, Arterburn JB, Kiselyov AS, Parker MA, Tkachenko SE, Savchuck NP, Sklar LA, Oprea TI, Prossnitz ER (2006) Virtual and biomolecular screening converge on a selective agonist for GPR30. Nat Chem Biol 2:207–212
Holm A, Baldetorp B, Olde B, Leeb-Lundberg LM, Nilsson BO (2011) The GPER1 agonist G-1 attenuates endothelial cell proliferation by inhibiting DNA synthesis and accumulating cells in the S and G2 phases of the cell cycle. J Vasc Res 48:327–335
Schwartz EL (2009) Antivascular actions of microtubule-binding drugs. Clin Cancer Res 15:2594–2601
Bevers LM, Braam B, Post JA, van Zonneveld AJ, Rabelink TJ, Koomans HS, Verhaar MC, Joles JA (2006) Tetrahydrobiopterin, but not l-arginine, decreases NO synthase uncoupling in cells expressing high levels of endothelial NO synthase. Hypertension 47:87–94
Odenlund M, Ekblad E, Nilsson BO (2008) Stimulation of oestrogen receptor-expressing endothelial cells with oestrogen reduces proliferation of cocultured vascular smooth muscle cells. Clin Exp Pharmacol Physiol 35:245–248
Andersson C, Lydrup ML, Ferno M, Idvall I, Gustafsson JA, Nilsson BO (2001) Immunocytochemical demonstration of oestrogen receptor β in blood vessels of the female rat. J Endocrinol 169:241–247
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Fellous A, Francon J, Lennon AM, Nunez J (1977) Microtubule assembly in vitro: purification of assembly-promoting factors. Eur J Biochem 78:167–174
Olmsted JB, Borisy GG (1973) Characterization of microtubule assembly in porcine brain extracts by viscometry. Biochemistry 12:4282–4289
Schachterle GR, Pollack RL (1973) A simplified method for the quantitative assay of small amounts of protein in biologic material. Anal Biochem 51:654–655
Eilken HM, Adams RH (2010) Dynamics of endothelial cell behavior in sprouting angiogenesis. Curr Opin Cell Biol 22:617–625
Batonnet S, Leibovitch MP, Tintignac L, Leibovitch SA (2004) Critical role for lysine 133 in the nuclear ubiquitin-mediated degradation of MyoD. J Biol Chem 279:5413–5420
Wallin M, Deinum J, Fridén B (1985) Interaction of estramustine phosphate with microtubule-associated proteins. FEBS Lett 179:289–293
Dhamodharan R, Jordan MA, Thrower D, Wilson L, Wadsworth P (1995) Vinblastine suppresses dynamics of individual microtubules in living interphase cells. Mol Biol Cell 6:1215–1229
Kreis T, Vale R (1999) Guidebook to the cytoskeletal and motor proteins. Oxford University Press, Oxford
Teng J, Wang ZY, Prossnitz ER, Bjorling DE (2008) The G protein-coupled receptor GPR30 inhibits human urothelial cell proliferation. Endocrinology 149:4024–4034
Acknowledgments
This study was supported by grants from the Swedish Research Council, the Swedish Dental Society, the Greta and Johan Kocks Foundation, the Sven and Lilly Thuréus Foundation and the Vascular Wall Programme at Lund University. We thank Dr. Mary Ann Jordan of the University of California at Santa Barbara for her thoughtful advice. Supported in part by a grant to RFL from the United States Department of Defence Breast Cancer Research Program W81XWH-10-1-0903. RFL and VP gratefully acknowledge the support of the Cancer Therapy and Research Center at the University of Texas Health Science Center San Antonio, an NCI-designated Cancer Center.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Holm, A., Grände, PO., Ludueña, R.F. et al. The G protein-coupled oestrogen receptor 1 agonist G-1 disrupts endothelial cell microtubule structure in a receptor-independent manner. Mol Cell Biochem 366, 239–249 (2012). https://doi.org/10.1007/s11010-012-1301-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-012-1301-3