Summary
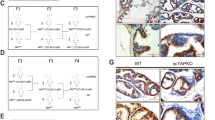
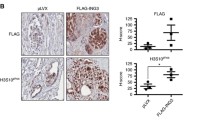
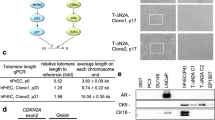
Hedgehog is a regulatory protein during embryonic development and its abnormal activation in adult tissues has been implicated in tumorigenesis within sites where epithelial–mesenchymal interactions take place. In the prostate, Hedgehog signaling activation was observed during advanced cancer progression and metastasis, but whether Hedgehog overexpression can initiate prostate tumorigenesis remains unknown. We introduced a Hedgehog-expressing vector by intra-prostate injection and electroporation to address the effects of Hedgehog overexpression. The manipulation caused lesions with characteristic prostatic intraepithelial neoplasia or even prostatic cancer (CaP) phenotypes within 30 days, with Hedgehog overexpression demonstrated by immunohistochemistry and Western blot detections. The tumorigenic phenotypes were confirmed by discontinuity of basal cell marker p63, mix-up of CK-8/CK-18 positive epithelial cells in the stoma as well as absence of α-SMA positive fibro-muscular sheath. Comparable Hedgehog overexpression was found in human CaP specimen. Thus, Hedgehog overexpression induced prostate tumorigenesis starting from the normal status. Furthermore, a mouse prostate cancer model induced by Hedgehog overexpression was established and may be used for testing novel therapeutical approaches targeting at Hedgehog signaling pathway.
Article PDF
Similar content being viewed by others
References
Greenlee R.T., Murray T., Bolden S. and Wingo P.A. (2000). Cancer statistics, 2000. CA Cancer J. Clin. 50: 7–33
Isaacs W., De Marzo A. and Nelson W.G. (2000). Focus on prostate cancer. Cancer Cell 2: 113–116
Abate-Shen C. and Shen M.M.(2000). Mouse models of prostate carcinogenesis. Trends Genet. 18: S1-S5
De Marzo A.M., Meeker A.K., Zha S., Luo J., Nakayama M., Platz E.A., Isaacs W.B. and Nelson W.G.(2003). Human prostate cancer precursors and pathobiology. Urology 62: 55–62
Huss W.J., Maddison L.A. and Greenberg N.M. (2001). Autochthonous mouse models for prostate cancer: past, present and future. Semin. Cancer Biol. 11: 245–260
Ingham P.W. and McMahon A.P. (2001). Hedgehog signaling in animal development: paradigms and principles. Genes Dev. 15: 3059–3087
McMahon A.P., Ingham P.W. and Tabin C.J.(2003). Developmental roles and clinical significance of hedgehog signaling. Curr. Top. Dev. Biol. 53:1–114
Podlasek C.A., Barnett D.H., Clemens J.Q., Bak P.M. and Bushman W.(1999). Prostate development requires Sonic hedgehog expressed by the urogenital sinus epithelium. Dev. Biol. 209: 28–39
Lamm M.L., Catbagan W.S., Laciak R.J., Barnett D.H., Hebner C.M., Gaffield W., Walterhouse D., Iannaccone P. and Bushman W. (2002). Sonic hedgehog activates mesenchymal Gli1 expression during prostate ductal bud formation. Dev. Biol. 249: 349–366
Freestone S.H., Marker P., Grace O.C., Tomlinson D.C., Cunha G.R., Harnden P. and Thomson A.A.(2003). Sonic hedgehog regulates prostatic growth and epithelial differentiation. Dev. Biol. 264: 352–362
Berman D.M., Desai N., Wang X., Karhadkar S.S., Reynon M., Abate-Shen C., Beachy P.A. and Shen M.M.(2004). Roles for Hedgehog signaling in androgen production and prostate ductal morphogenesis. Dev. Biol. 267: 387–398
Watkins D.N., Berman D.M., Burkholder S.G., Wang B., Beachy P.A. and Baylin S.B.(2003). Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 422: 313–317
Thayer S.P., di Magliano M.P., Heiser P.W., Nielsen C.M., Roberts D.J., Lauwers G.Y., Qi Y.P., Gysin S., Fernandez-del Castillo C., Yajnik V., Antoniu B., McMahon M., Warshaw A.L. and Hebrok M.(2003). Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 425: 851–856
Berman D.M., Karhadkar S.S., Maitra A., Montes De Oca R., Gerstenblith M.R., Briggs K., Parker A.R., Shimada Y., Eshleman J.R., Watkins D.N. and Beachy P.A. (2003). Widespread requirement for Hedgehog ligand stimulation in growth of digestive tract tumours. Nature 425: 846–851
Kumamoto H., Ohki K. and Ooya K.(2004). Expression of Sonic hedgehog (SHH) signaling molecules in ameloblastomas. J. Oral Pathol. Med. 33: 185–190
Goodrich L.V., Milenkovic L., Higgins K.M. and Scott M.P.(1997). Altered neural cell fates and medulloblastoma in mouse patched mutants. Science 277: 1109–1113
Oro A.E., Higgins K.M., Hu Z., Bonifas J.M., Epstein E.H., Jr. and Scott M.P.(1997). Basal cell carcinomas in mice overexpressing sonic hedgehog. Science 276: 817–821
Aboulkassim T.O., LaRue H., Lemieux P., Rousseau F. and Fradet Y. (2003). Alteration of the PATCHED locus in superficial bladder cancer. Oncogene 22: 2967–2971
Dahmane N., Sanchez P., Gitton Y., Palma V., Sun T., Beyna M., Weiner H. and Ruiz i Altaba A. (2001). The Sonic Hedgehog-Gli pathway regulates dorsal brain growth and tumorigenesis. Development 128: 5201–5212
Sanchez P., Hernandez A.M., Stecca B., Kahler A.J., DeGueme A.M., Barrett A., Beyna M., Datta M.W., Datta S. and Ruiz i Altaba A.(2004). Inhibition of prostate cancer proliferation by interference with SONIC HEDGEHOG-GLI1 signaling. Proc. Natl. Acad. Sci. USA 101: 12561–12566
Sheng T., Li C., Zhang X., Chi S., He N., Chen K., McCormick F., Gatalica Z. and Xie J.(2004). Activation of the hedgehog pathway in advanced prostate cancer. Mol. Cancer 3: 29
Fan L., Pepicelli C.V., Dibble C.C., Catbagan W., Zarycki J.L., Laciak R., Gipp J., Shaw A., Lamm M.L., Munoz A. Lipinski R., Thrasher J.B. and Bushman W. (2004) Hedgehog signaling promotes prostate xenograft tumor growth. Endocrinology 145: 3961–3970
Karhadkar S.S., Bova G.S., Abdallah N., Dhara S., Gardner D., Maitra A., Isaacs J.T., Berman D.M. and Beachy P.A. (2004). Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature 431: 707–712
Oberg K.C., Pira C.U., Revelli J.P., Ratz B., Aguilar-Cordova E. and Eichele G.(2002). Efficient ectopic gene expression targeting chick mesoderm. Dev. Dyn. 224: 291–302
Nakamura H. and Funahashi J.(2001). Introduction of DNA into chick embryos by in ovo electroporation. Methods 24: 43–48
Park J.H., Walls J.E., Galvez J.J., Kim M., Abate-Shen C., Shen M.M. and Cardiff R.D. (2002). Prostatic intraepithelial neoplasia in genetically engineered mice. Am. J. Pathol. 161: 727–735
Peterson G.L.(1997). A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal. Biochem. 83: 346–356
Laemmli U.K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Yang J., Mani S.A., Donaher J.L., Ramaswamy S., Itzykson R.A., Come C., Savagner P., Gitelman I., Richardson A. and Weinberg R.A.(2004). Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 117: 927–939
Kwok W.K., Ling M.T., Lee T.W., Lau T.C., Zhou C., Zhang X., Chua C.W., Chan K.W., Chan F.L., Glackin C., Wong Y.C. and Wang X.(2005). Up-regulation of TWIST in prostate cancer and its implication as a therapeutic target. Cancer Res. 65: 5153–5162
Abate-Shen C. and Shen M.M. (2000). Molecular genetics of prostate cancer. Genes Dev. 14: 2410–2434
van Leenders G.J. and Schalken J.A. (2003). Epithelial cell differentiation in the human prostate epithelium: implications for the pathogenesis and therapy of prostate cancer. Crit. Rev. Oncol. Hematol. 46: S3-S10
Sung S.Y. and Chung L.W.(2002) Prostate tumor–stroma interaction: molecular mechanisms and opportunities for therapeutic targeting. Differentiation 70: 506–521
Wong Y.C. and Tam N.N.(2002). Dedifferentiation of stromal smooth muscle as a factor in prostate carcinogenesis. Differentiation 70: 633–645
Fudge K., Bostwick D.G. and Stearns M.E. (1996). Platelet-derived growth factor A and B chains and the alpha and beta receptors in prostatic intraepithelial neoplasia. Prostate 29: 282–286
Kaplan P.J., Mohan S., Cohen P., Foster B.A. and Greenberg N.M. (1999). The insulin-like growth factor axis and prostate cancer: lessons from the transgenic adenocarcinoma of mouse prostate (TRAMP) model. Cancer Res. 59: 2203–2209
Jin C., McKeehan K., Guo W., Jauma S., Ittmann M.M., Foster B., Greenberg N.M., McKeehan W.L. and Wang F. (2003). Cooperation between ectopic FGFR1 and depression of FGFR2 in induction of prostatic intraepithelial neoplasia in the mouse prostate. Cancer Res. 63: 8784–8790
Evangelou A.I., Winter S.F., Huss W.J., Bok R.A. and Greenberg N.M.(2004) Steroid hormones, polypeptide growth factors, hormone refractory prostate cancer, and the neuroendocrine phenotype. J. Cell. Biochem. 91: 671–683
Rice R., Spencer-Dene B., Connor E.C., Gritli-Linde A., McMahon A.P., Dickson C., Thesleff I. and Rice D.P.(2004) Disruption of Fgf10/Fgfr2b-coordinated epithelial-mesenchymal interactions causes cleft palate. J. Clin. Invest. 113: 1692–1700
Navone N.M., Logothetis C.J., von Eschenbach A.C. and Troncoso P.(1998). Model systems of prostate cancer: uses and limitations. Cancer Metast. Rev. 17: 361–371
Huss W.J., Hanrahan C.F., Barrios R.J., Simons J.W. and Greenberg N.M.(2001). Angiogenesis and prostate cancer: identification of a molecular progression switch. Cancer Res. 61: 2736–2743
Masumori N., Tsuchiya K., Tu W.H., Lee C., Kasper S., Tsukamoto T., Shappell S.B. and Matusik R.J. (2004). An allograft model of androgen independent prostatic neuroendocrine carcinoma derived from a large probasin promoter-T antigen transgenic mouse line. J. Urol. 171: 439–442
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, BY., Lin, D.PC., Liu, JY. et al. A mouse prostate cancer model induced by Hedgehog overexpression. J Biomed Sci 13, 373–384 (2006). https://doi.org/10.1007/s11373-005-9050-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11373-005-9050-x