Abstract
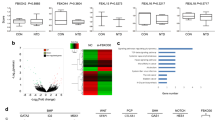
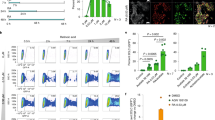
RETINOIDS (vitamin A and its metabolites) are suspected of regulating diverse aspects of growth, differentiation, and patterning during embryogenesis1, but many questions remain about the identities and functions of the endogenous active retinoids involved. The pleiotropic effects of retinoids may be explained by the existence of complex signal transduction pathways involving diverse nuclear receptors of the retinoic acid receptor (RAR) and retinoid X receptor (RXR) families, and at least two types of cellular retinoic acid binding proteins (CRABP-I and -II)2. The different RARs, RXRs, and CRABPs have different expression patterns during vertebrate embryogenesis2,3, suggesting that they each have particular functions. Another level at which fine tuning of retinoid action could occur is the metabolism of vitamin A to active metabolites, which may include all-trans-retinoic acid4–7, all-trans-3,4-didehydroretinoic acid8, 9-cis-retinoic acid9,10, and 14-hydroxy-4,14-retroretinol11. Formation of the metabolite all-trans-4-oxo-retinoie acid from retinoic acid was considered to be an inactivation pathway during growth and differentiation12–14. We report here that, in contrast, 4-oxo-retinoic acid is a highly active metabolite which can modulate positional specification in early embryos. We also show that this retinoid binds avidly to and activates RAR& beta;, and that it is available in early embryos. The different activities of 4-oxo-retinoic acid and retinoic acid in modulating positional specification on the one hand, and growth and differentiation on the other, interest us in the possibility that specific retinoid ligands regulate different physiological processes in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Maden, M. & Holder, H. Bioessays 14, 431–438 (1992).
Leid, M., Kastner, P. & Chambon, P. Trends Biochem. Sci. 17, 427–433 (1992).
Ellinger-Ziegelbauer, H. & Dreyer, C. Genes Dev. 5, 94–104 (1991).
Tickle, C., Alberts, B., Wolpert, L. & Lee, J. Nature 296, 564–566 (1982).
Thaller, C. & Eichele, G. Nature 327, 625–628 (1987).
Durston, A. J. et al. Nature 340, 140–144 (1989).
Hogan, B. L. M., Thaller, C. & Eichele, G. Nature 359, 237–241 (1992).
Thaller, C. & Eichele, G. Nature 345, 815–819 (1990).
Levin, A. A. et al. Nature 355, 359–361 (1992).
Heyman, R. A. et al. Cell 68, 397–406 (1992).
Buck, J. et al. Science 254, 1654–1656 (1991).
Frolik, C. A. et al. Biochemistry 18, 2092–2097 (1979).
Surekha Rao, M. S., John, J. & Cama, H. R. Int. J. Vitam. Nutr. Res. 42, 368–378 (1972).
Williams, J. B., Shoelds, C. O., Brettel, L. M. & Napoli, J. L. Analyt. Biochem. 160, 267–274 (1987).
Sive, H. L., Draper, B. W., Harland, R. M. & Weintraub, H. Genes Dev. 4, 932–942 (1990).
Ruiz i Altaba, A. & Jessell, T. Genes Dev. 5, 175–187 (1991).
Wilkinson, D. G. et al. Nature 341, 405–409 (1989).
Simeone, A. et al. Nature 346, 763–766 (1990).
Dekker, E. J. et al. Mech. Dev. 40, 3–12 (1992).
Eckhoff, C. & Nau, H. J. Lipid Res. 31, 1445–1454 (1990).
Allenby, G. et al. Proc. natn. Acad. Sci. U.S.A. 90, 30–34 (1993).
Nagpal, S. et al. Cell 70, 1007–1019 (1992).
Folkers, G. E., van der Leede, B. M. & van der Saag, P. T. Molec. Endocr. 7, 616–627 (1993).
Sani, B. P. in Retinoids (eds Livrea, M. A. & Packer, L.) 237–247 (Marcel Dekker, New York, 1993).
Frolik, C. A., Roller, P. P., Roberts, A. B. & Sporn, M. B. J. biol. Chem. 255, 8057–8062 (1980).
Roberts, A. B., Lamb, L. C. & Sporn, M. B. Arch. Biochem. Biophys. 199, 374–383 (1980).
Fiorella, P. D. & Napoli, J. L. J. biol. Chem. 266, 16572–16579 (1991).
Nieuwkoop, P. D. et al. J. exp. Zool. 120, 1–108 (1952).
Kao, K. R. & Elinson, R. P. Devl Biol. 127, 64–77 (1988).
Palmer, F. B. St. C. Biochim. biophys. Acta 231, 134–144 (1971).
Handyside, A. H., O'Neill, G. T., Jones, M. & Hooper, M. L. Roux's Arch. dev. Biol. 198, 48–55 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pijnappel, W., Hendriks, H., Folkers, G. et al. The retinoid ligand 4-oxo-retinoic acid is a highly active modulator of positional specification. Nature 366, 340–344 (1993). https://doi.org/10.1038/366340a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/366340a0
This article is cited by
-
Genetics and functions of the retinoic acid pathway, with special emphasis on the eye
Human Genomics (2019)
-
Genetic architecture of retinoic-acid signaling-associated ocular developmental defects
Human Genetics (2019)
-
Identification of influential proteins in the classical retinoic acid signaling pathway
Theoretical Biology and Medical Modelling (2018)
-
Neurogenesis in Response to Synthetic Retinoids at Different Temporal Scales
Molecular Neurobiology (2018)
-
Chemical activation of RARβ induces post-embryonically bilateral limb duplication during Xenopus limb regeneration
Scientific Reports (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.