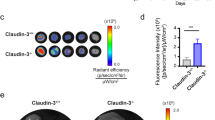
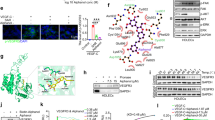
Abstract
Metastasis to local lymph nodes via the lymphatic vessels is a common step in the spread of solid tumors. To investigate the molecular mechanisms underlying the spread of cancer by the lymphatics, we examined the ability of vascular endothelial growth factor (VEGF)-D, a ligand for the lymphatic growth factor receptor VEGFR-3/Flt-4, to induce formation of lymphatics in a mouse tumor model. Staining with markers specific for lymphatic endothelium demonstrated that VEGF-D induced the formation of lymphatics within tumors. Moreover, expression of VEGF-D in tumor cells led to spread of the tumor to lymph nodes, whereas expression of VEGF, an angiogenic growth factor which activates VEGFR-2 but not VEGFR-3, did not. VEGF-D also promoted tumor angiogenesis and growth. Lymphatic spread induced by VEGF-D could be blocked with an antibody specific for VEGF-D. This study demonstrates that lymphatics can be established in solid tumors and implicates VEGF family members in determining the route of metastatic spread.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Liotta, L.A. Cancer cell invasion and metastasis. Sci. Am. 266, 34–41 (1992).
O'Reilly, M.S. et al. Angiostatin: a novel angiogenesis inhibitor that mediates the supression of metastases by a lewis lung carcinoma. Cell 79, 315–328 (1994).
O'Reilly, M.S. et al. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88, 277–285 (1997).
Millauer, B., Shawver, L.K., Plate, K.H., Risau, W. & Ullrich, A. Glioblastoma growth inhibited in vivo by a dominant negative Flk-1 mutant. Nature 367, 576–579 (1994).
Millauer, B. et al. Dominant-negative inhibition of Flk-1 suppresses the growth of many tumor types in vivo. Cancer Res. 56, 1615–1620 (1996).
Kim, K.J. et al. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 362, 841–844 (1993).
Saleh, M., Stacker, S.A. & Wilks, A.F. Inhibition of growth of C6 glioma cells in vivo by expression of antisense vascular endothelial growth factor sequence. Cancer Res. 56, 393–401 (1996).
Achen, M.G. et al. Vascular endothelial growth factor D (VEGF-D) is a ligand for the tyrosine kinases VEGF receptor 2 (Flk-1) and VEGF receptor 3 (Flt-4). Proc. Natl. Acad. Sci. USA 95, 548–553 (1998).
Orlandini, M., Marconcini, L., Ferruzzi, R. & Oliviero, S. Identification of a C-Fos-induced gene that is related to the platelet-derived growth factor/vascular endothelial growth factor family. Proc. Natl. Acad. Sci. USA 93, 11675–11680 (1996).
Kaipainen, A. et al. Expression of the Fms-like tyrosine kinase 4 gene becomes restricted to lymphatic endothelium during development. Proc. Natl. Acad. Sci. USA 92, 3566–3570 (1995).
Taipale, J. et al. Vascular endothelial growth factor receptor-3. Curr. Top. Microbiol. Immunol. 237, 85–96 (1999).
Dukes, C.E. The classification of cancer of the rectum. J. Pathol. 35, 323–332 (1932).
Fisher, B. et al. Relation of number of positive axillary nodes to the prognosis of patients with primary breast cancer. An NSABP update. Cancer 52, 1551–1557 (1983).
Stacker, S.A. & Achen, M.G. The vascular endothelial growth factor family: signaling for vascular development. Growth Factors 17, 1–11 (1999).
Stacker, S.A. et al. Biosynthesis of vascular endothelial growth factor-D involves proteolytic processing which generates non-covalent homodimers. J. Biol. Chem. 274, 32127–32136 (1999).
Achen, M.G. et al. Monoclonal antibodies to vascular endothelial growth factor-D block interactions with both VEGF receptor-2 and VEGF receptor-3. Eur.J.Biochem. 267, 2505–2515 (2000).
Valtola, R. et al. VEGFR-3 and its ligand VEGF-C are associated with angiogenesis in breast cancer. Am. J. Pathol. 154, 1381–1390 (1999).
Banerji, S. et al. LYVE-1, a new homologue of the CD44 glycoprotein, is a lymph-specific receptor for hyaluronan. J. Cell Biol. 144, 789–801 (1999).
de Waal, R.M.W. et al. Lack of lymphangiogenesis in human primary cutaneous melanoma. Consequences for the mechanism of lymphatic dissemination. Am. J. Pathol. 150, 1951–1957 (1997).
Prewett, M. et al. Antivascular endothelial growth factor receptor (fetal liver kinase 1) monoclonal antibody inhibits tumor angiogenesis and growth of several mouse and human tumors. Cancer Res. 59, 5209–5218 (1999).
Whiteside, T.L. & Herberman, R.B. The role of natural killer cells in immune surveillance of cancer. Curr. Opin. Immunol. 7, 704–710 (1995).
Vujanovic, N.L., Basse, P., Herberman, R.B. & Whiteside, T.L. Antitumor functions of natural killer cells and control of metastases. Methods 9, 394–408 (1996).
Babu, S., Porte, P., Klei, T.R., Shultz, L.D. & Rajan, T.V. Host NK cells are required for the growth of the human filarial parasite Brugia malayi in mice. J. Immunol. 161, 1428–1432 (1998).
Ansink, A.C. et al. Identification of sentinel lymph nodes in vulvar carcinoma patients with the aid of a Patent Blue V injection: a multicenter study. Cancer 86, 652–656 (1999).
Folkman, J. Anti-angiogenesis: new concepts of therapy of solid tumors. Ann. Surg. 175, 409–416 (1972).
Folkman, J. & Klagsburn, M. Angiogenic factors. Science 235, 442–447 (1987).
Folkman, J. What is the evidence that tumors are angiogenesis dependent? J. Natl. Cancer Inst. 82, 4–6 (1990).
Folkman, J. & D'Amore, P.A. Blood vessel formation:what is its molecular basis? Cell 87, 1153–1155 (1996).
Wood, J.M. et al. PTK787/ZK 222584, a novel and potent inhibitor of vascular endothelial growth factor receptor tyrosine kinases, impairs vascular endothelial growth factor-induced responses and tumor growth after oral administration. Cancer Res. 60, 2178–2189 (2000).
Drevs, J. et al. Effects of PTK787/ZK 222584, a specific inhibitor of vascular endothelial growth factor receptor tyrosine kinases, on primary tumor, metastasis, vessel density, and blood flow in a murine renal cell carcinoma model. Cancer Res. 60, 4819–4824 (2000).
Lymboussaki, A., Achen, M.G., Stacker, S.A. & Alitalo, K. Growth factors regulating lymphatic vessels. Curr. Top. Microbiol. Immunol. 251, 75–82 (2000)
Leu, A.J., Berk, D.A., Lymboussaki, A., Alitalo, K. & Jain, R.K. Absence of functional lymphatics within a murine sarcoma: a molecular and functional evaluation. Cancer Res. 60, 4324–4327 (2000).
Lymboussaki, A. et al. Expression of the vascular endothelial growth factor C receptor VEGFR-3 in lymphatic endothelium of the skin and in vascular tumors. Am. J. Pathol. 153, 395–403 (1998).
Joukov, V. et al. A novel vascular endothelial growth factor, VEGF-C, Is a ligand for the Flt-4 (VEGFR-3) and KDR (VEGFR-2) receptor tyrosine kinases. EMBO J. 15, 290–298 (1996).
Kubo, H. et al. Involvement of vascular endothelial growth factor receptor-3 in maintenance of integrity of endothelial cell lining during tumor angiogenesis. Blood 96, 546–553 (2000).
Salven, P. et al. Vascular endothelial growth factors VEGF-B and VEGF-C are expressed in human tumors. Am. J. Pathol. 153, 103–108 (1998).
Achen, M.G. et al. Localisation of vascular endothelial growth factor-D in malignant melanoma suggests a role in tumour angiogenesis J. Pathol. (in the press).
Plate, K.H., Breier, G., Millauer, B., Ullrich, A. & Risau, W. Up-regulation of vascular endothelial growth factor and its cognate receptors in a rat glioma model of tumor angiogenesis. Cancer Res. 53, 5822–5827 (1993).
Niki, T. et al. Expression of vascular endothelial growth factors A, B, C, and D and their relationships to lymph node status in lung adenocarcinoma. Clin. Cancer Res. 6, 2431–2439 (2000).
Mandriota, S.J. et al. Vascular endothelial growth factor-C-mediated lymphangiogenesis promotes tumor metastasis. EMBOJ. in press (2001).
Layton, J.E., Iaria, J. & Nicholson, S.E. Neutralising antibodies to the granulocyte colony-stimulating factor receptor recognise both the immunoglobulin-like domain and the cytokine receptor homologous domain. Growth Factors. 14, 117–130 (1997).
Acknowledgements
We thank T. Helman and the staff of the animal house at the Ludwig Institute for Cancer Research for assistance with the SCID mice; A. Scott and C. Hall for advice on animal tumor models; B. Mann for discussions on dye injection; M. Smyth for discussions on mouse models; S. Squinto for supplying the pAPEX vector; and A. Burgess and M. Hibbs for critical reading of this manuscript. This work was funded in part by project grants from the National Health and Medical Research Council of Australia and the Anti-Cancer Council of Victoria. D. J. is supported by the UK Medical Research Council and by a project grant (00-311) from the Association for International Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stacker, S., Caesar, C., Baldwin, M. et al. VEGF-D promotes the metastatic spread of tumor cells via the lymphatics. Nat Med 7, 186–191 (2001). https://doi.org/10.1038/84635
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/84635
This article is cited by
-
Associations amongst genes, molecules, cells, and organs in breast cancer metastasis
Clinical & Experimental Metastasis (2023)
-
Tailoring Treatment for Patients with Inflammatory Breast Cancer
Current Treatment Options in Oncology (2023)
-
Identification of Differentially Expressed Genes in Human Colorectal Cancer Using RNASeq Data Validated on the Molecular Level with Real-Time PCR
Biochemical Genetics (2023)
-
The lymphatic system and sentinel lymph nodes: conduit for cancer metastasis
Clinical & Experimental Metastasis (2022)
-
FABP4 is an independent risk factor for lymph node metastasis and poor prognosis in patients with cervical cancer
Cancer Cell International (2021)