Abstract
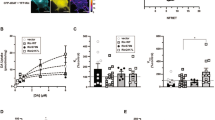
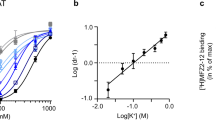
Phosphatidylinositol (4,5)-bisphosphate (PIP2) regulates the function of ion channels and transporters. Here, we demonstrate that PIP2 directly binds the human dopamine (DA) transporter (hDAT), a key regulator of DA homeostasis and a target of the psychostimulant amphetamine (AMPH). This binding occurs through electrostatic interactions with positively charged hDAT N-terminal residues and is shown to facilitate AMPH-induced, DAT-mediated DA efflux and the psychomotor properties of AMPH. Substitution of these residues with uncharged amino acids reduces hDAT-PIP2 interactions and AMPH-induced DA efflux without altering the hDAT physiological function of DA uptake. We evaluated the significance of this interaction in vivo using locomotion as a behavioral assay in Drosophila melanogaster. Expression of mutated hDAT with reduced PIP2 interaction in Drosophila DA neurons impairs AMPH-induced locomotion without altering basal locomotion. We present what is to our knowledge the first demonstration of how PIP2 interactions with a membrane protein can regulate the behaviors of complex organisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Suh, B.C. & Hille, B. PIP2 is a necessary cofactor for ion channel function: how and why? Annu Rev Biophys 37, 175–195 (2008).
McLaughlin, S. & Murray, D. Plasma membrane phosphoinositide organization by protein electrostatics. Nature 438, 605–611 (2005).
Kadamur, G. & Ross, E.M. Mammalian phospholipase C. Annu. Rev. Physiol. 75, 127–154 (2013).
Czech, M.P. PIP2 and PIP3: complex roles at the cell surface. Cell 100, 603–606 (2000).
Ben-Aissa, K. et al. Activation of moesin, a protein that links actin cytoskeleton to the plasma membrane, occurs by phosphatidylinositol 4,5-bisphosphate (PIP2) binding sequentially to two sites and releasing an autoinhibitory linker. J. Biol. Chem. 287, 16311–16323 (2012).
Whorton, M.R. & MacKinnon, R. Crystal structure of the mammalian GIRK2 K+ channel and gating regulation by G proteins, PIP2, and sodium. Cell 147, 199–208 (2011).
Buchmayer, F. et al. Amphetamine actions at the serotonin transporter rely on the availability of phosphatidylinositol-4,5-bisphosphate. Proc. Natl. Acad. Sci. USA 110, 11642–11647 (2013).
Pizzo, A.B. et al. The membrane raft protein Flotillin-1 is essential in dopamine neurons for amphetamine-induced behavior in Drosophila. Mol. Psychiatry 18, 824–833 (2013).
Hamilton, P.J. et al. De novo mutation in the dopamine transporter gene associates dopamine dysfunction with autism spectrum disorder. Mol. Psychiatry 18, 1315–1353 (2013).
Giros, B., Jaber, M., Jones, S.R., Wightman, R.M. & Caron, M.G. Hyperlocomotion and indifference to cocaine and amphetamine in mice lacking the dopamine transporter. Nature 379, 606–612 (1996).
Sulzer, D., Sonders, M.S., Poulsen, N.W. & Galli, A. Mechanisms of neurotransmitter release by amphetamines: a review. Prog. Neurobiol. 75, 406–433 (2005).
Robertson, S.D., Matthies, H.J. & Galli, A. A closer look at amphetamine-induced reverse transport and trafficking of the dopamine and norepinephrine transporters. Mol. Neurobiol. 39, 73–80 (2009).
Cervinski, M.A., Foster, J.D. & Vaughan, R.A. Psychoactive substrates stimulate dopamine transporter phosphorylation and down regulation by cocaine sensitive and protein kinase C dependent mechanisms. J. Biol. Chem. 280, 40442–40449 (2005).
Khoshbouei, H. et al. N-terminal phosphorylation of the dopamine transporter is required for amphetamine-induced efflux. PLoS Biol. 2, E78 (2004).
Fog, J.U. et al. Calmodulin kinase II interacts with the dopamine transporter C terminus to regulate amphetamine-induced reverse transport. Neuron 51, 417–429 (2006).
Cremona, M.L. et al. Flotillin-1 is essential for PKC-triggered endocytosis and membrane microdomain localization of DAT. Nat. Neurosci. 14, 469–477 (2011).
Wang, J. & Richards, D.A. Segregation of PIP2 and PIP3 into distinct nanoscale regions within the plasma membrane. Biol. Open 1, 857–862 (2012).
Hope, H.R. & Pike, L.J. Phosphoinositides and phosphoinositide-utilizing enzymes in detergent-insoluble lipid domains. Mol. Biol. Cell 7, 843–851 (1996).
Várnai, P. & Balla, T. Visualization of phosphoinositides that bind pleckstrin homology domains: calcium- and agonist-induced dynamic changes and relationship to myo-[3H]inositol-labeled phosphoinositide pools. J. Cell Biol. 143, 501–510 (1998).
McLaughlin, S., Wang, J., Gambhir, A. & Murray, D. PIP2 and proteins: interactions, organization, and information flow. Annu. Rev. Biophys. Biomol. Struct. 31, 151–175 (2002).
Hilgemann, D.W. & Ball, R. Regulation of cardiac Na+, Ca2+ exchange and KATP potassium channels by PIP2 . Science 273, 956–959 (1996).
Bowton, E. et al. Dysregulation of dopamine transporters via dopamine D2 autoreceptors triggers anomalous dopamine efflux associated with attention-deficit hyperactivity disorder. J. Neurosci. 30, 6048–6057 (2010).
Binda, F. et al. Syntaxin 1A interaction with the dopamine transporter promotes amphetamine-induced dopamine efflux. Mol. Pharmacol. 74, 1101–1108 (2008).
Várnai, P. et al. Inositol lipid binding and membrane localization of isolated pleckstrin homology (PH) domains. Studies on the PH domains of phospholipase C δ1 and p130. J. Biol. Chem. 277, 27412–27422 (2002).
Robbins, J., Marsh, S.J. & Brown, D.A. Probing the regulation of M (Kv7) potassium channels in intact neurons with membrane-targeted peptides. J. Neurosci. 26, 7950–7961 (2006).
Beuming, T. et al. The binding sites for cocaine and dopamine in the dopamine transporter overlap. Nat. Neurosci. 11, 780–789 (2008).
Bisgaard, H. et al. The binding sites for benztropines and dopamine in the dopamine transporter overlap. Neuropharmacology 60, 182–190 (2011).
Kniazeff, J. et al. An intracellular interaction network regulates conformational transitions in the dopamine transporter. J. Biol. Chem. 283, 17691–17701 (2008).
Khelashvili, G., Galli, A. & Weinstein, H. Phosphatidylinositol 4,5-biphosphate (PIP2) lipids regulate the phosphorylation of syntaxin N-terminus by modulating both its position and local structure. Biochemistry 51, 7685–7698 (2012).
Khelashvili, G., Harries, D. & Weinstein, H. Modeling membrane deformations and lipid demixing upon protein-membrane interaction: the BAR dimer adsorption. Biophys. J. 97, 1626–1635 (2009).
Khelashvili, G., Weinstein, H. & Harries, D. Protein diffusion on charged membranes: a dynamic mean-field model describes time evolution and lipid reorganization. Biophys. J. 94, 2580–2597 (2008).
Balla, T., Szentpetery, Z. & Kim, Y.J. Phosphoinositide signaling: new tools and insights. Physiology (Bethesda) 24, 231–244 (2009).
Wicker-Thomas, C. & Hamann, M. Interaction of dopamine, female pheromones, locomotion and sex behavior in Drosophila melanogaster. J. Insect Physiol. 54, 1423–1431 (2008).
Pendleton, R.G., Rasheed, A., Sardina, T., Tully, T. & Hillman, R. Effects of tyrosine hydroxylase mutants on locomotor activity in Drosophila: a study in functional genomics. Behav. Genet. 32, 89–94 (2002).
Kume, K., Kume, S., Park, S.K., Hirsh, J. & Jackson, F.R. Dopamine is a regulator of arousal in the fruit fly. J. Neurosci. 25, 7377–7384 (2005).
van Rheenen, J., Achame, E.M., Janssen, H., Calafat, J. & Jalink, K. PIP2 signaling in lipid domains: a critical re-evaluation. EMBO J. 24, 1664–1673 (2005).
Sorkina, T., Caltagarone, J. & Sorkin, A. Flotillins regulate membrane mobility of the dopamine transporter but are not required for its protein kinase C dependent endocytosis. Traffic 14, 709–724 (2013).
Gabriel, L.R. et al. Dopamine transporter endocytic trafficking in striatal dopaminergic neurons: differential dependence on dynamin and the actin cytoskeleton. J. Neurosci. 33, 17836–17846 (2013).
Mazei-Robison, M.S. et al. Anomalous dopamine release associated with a human dopamine transporter coding variant. J. Neurosci. 28, 7040–7046 (2008).
Das, R. & Baker, D. Macromolecular modeling with Rosetta. Annu. Rev. Biochem. 77, 363–382 (2008).
Gracia, L. RMSDTT: RMSD Trajectory Tool, 2.5 ed., (Weill Medical College of Cornell University, Department of Physiology and Biophysics, 2005).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–8, 27–8 (1996).
Earl, D.J. & Deem, M.W. Parallel tempering: theory, applications, and new perspectives. Phys. Chem. Chem. Phys. 7, 3910–3916 (2005).
Kiessling, V., Wan, C. & Tamm, L.K. Domain coupling in asymmetric lipid bilayers. Biochim. Biophys. Acta 1788, 64–71 (2009).
Šali, A. & Blundell, T.L. Comparative protein modelling by satisfaction of spatial restraints. J. Mol. Biol. 234, 779–815 (1993).
Sharp, K.A. & Honig, B. Electrostatic interactions in macromolecules: theory and applications. Annu. Rev. Biophys. Biophys. Chem. 19, 301–332 (1990).
Chaikin, P.M. & Lubensky, T.C. Principles of Condensed Matter Physics (Cambridge University Press, Cambridge, 2000).
Mackerell, A.D. Jr., Feig, M. & Brooks, C.L. III. Extending the treatment of backbone energetics in protein force fields: limitations of gas-phase quantum mechanics in reproducing protein conformational distributions in molecular dynamics simulations. J. Comput. Chem. 25, 1400–1415 (2004).
Friggi-Grelin, F. et al. Targeted gene expression in Drosophila dopaminergic cells using regulatory sequences from tyrosine hydroxylase. J. Neurobiol. 54, 618–627 (2003).
Wang, J.W., Beck, E.S. & McCabe, B.D. A modular toolset for recombination transgenesis and neurogenetic analysis of Drosophila. PLoS ONE 7, e42102 (2012).
Acknowledgements
We thank S. Singh (Yale University) for the gift of the dDAT cDNA. Computational resources were provided by Teragrid allocation MCB120008 on the Ranger machine, by the NERSC allocation (repository m1710) on the Carver supercomputer and the David Cofrin Center for Biomedical Information of the Institute for Computational Biomedicine at Weill Cornell Medical College. This work was supported by US National Science Foundation Graduate Research Fellowship DGE0909667 (P.J.H.) and grants F31 DA 035535-01 (P.J.H.), 23658-B11 (H.H.S.), DA035263 (A.G.), P01 DA012408 (A.G., H.W. and J.A.J.) and U54GM087519 (H.W. and J.A.J.).
Author information
Authors and Affiliations
Contributions
P.J.H., A.N.B., G.K., C.S., K.E. and H.J.G.M. performed experiments. P.J.H., A.N.B., G.K., C.S., K.E., J.A.J., H.H.S., H.W., H.J.G.M. and A.G. designed experiments and analyzed data. G.K. and H.W. generated computational models and analyses. J.A.J. supplied expression vectors and plasmid DNA. P.J.H., A.N.B., H.W., H.J.G.M., G.K. and A.G. wrote the manuscript. All of the authors participated in the discussion of results and contributed to the preparation of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results and Supplementary Figures 1–11. (PDF 955 kb)
Rights and permissions
About this article
Cite this article
Hamilton, P., Belovich, A., Khelashvili, G. et al. PIP2 regulates psychostimulant behaviors through its interaction with a membrane protein. Nat Chem Biol 10, 582–589 (2014). https://doi.org/10.1038/nchembio.1545
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1545
This article is cited by
-
Electrostatic switch mechanisms of membrane protein trafficking and regulation
Biophysical Reviews (2023)
-
Lipid-mediated prestin organization in outer hair cell membranes and its implications in sound amplification
Nature Communications (2022)
-
The Role of the Dopamine Transporter in the Effects of Amphetamine on Sleep and Sleep Architecture in Drosophila
Neurochemical Research (2022)
-
Phosphatidylinositol 4,5-bisphosphate (PIP2) facilitates norepinephrine transporter dimerization and modulates substrate efflux
Communications Biology (2022)
-
A network of phosphatidylinositol (4,5)-bisphosphate (PIP2) binding sites on the dopamine transporter regulates amphetamine behavior in Drosophila Melanogaster
Molecular Psychiatry (2021)