Abstract
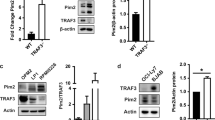
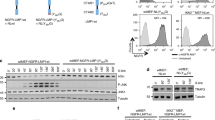
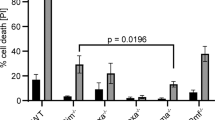
The c-Jun NH2-terminal kinase (JNK) is implicated in the apoptotic response of cells exposed to stress1, but the JNK signal transduction pathway may not act exclusively in apoptosis1. In some studies of tumor cells, JNK has been implicated in signaling cell survival1. The possibility that JNK might mediate a survival signal in tumor cells is consistent with the observation that it is activated in response to some oncogenes, such as the leukemogenic oncogene BCR–ABL, which is created by a reciprocal translocation between human chromosomes 9 and 22 (ref. 2). The BCR-ABL protein activates the JNK signaling pathway in hematopoietic cells and increases transcriptional activity mediated by the transcription factor AP1 (ref. 3). Also, inhibition of c-Jun or JNK prevents BCR–ABL-induced cell transformation in vitro3,4. Although this implicates the JNK signaling pathway in transformation by BCR–ABL, the possible role of JNK in this process is unclear. We find that disruption of the JNK ortholog Mapk8 (also known as Jnk1) in mice causes defective transformation of pre-B cells by BCR–ABL in vitro and in vivo. The Jnk1 protein is required for the survival of the transformed cells in the absence of stromal support. Failure to survive is associated with decreased expression of Bcl2, and the effect of Jnk1 deficiency can be rescued by transgenic expression of Bcl2. Our results show that Jnk1 signals cell survival in transformed B lymphoblasts and suggest that it may contribute to the pathogenesis of some proliferative diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Davis, R.J. Signal transduction by the JNK group of MAP kinases. Cell 103, 239–252 (2000).
Sawyers, C.L. Chronic myeloid leukemia. N. Engl. J. Med. 340, 1330–1340 (1999).
Raitano, A.B., Halpern, J.R., Hambuch, T.M. & Sawyers, C.L. The Bcr-Abl leukemia oncogene activates Jun kinase and requires Jun for transformation. Proc. Natl Acad. Sci. USA 92, 11746–11750 (1995).
Dickens, M. et al. A cytoplasmic inhibitor of the JNK signal transduction pathway. Science 277, 693–696 (1997).
McLaughlin, J., Chianese, E. & Witte, O.N. In vitro transformation of immature hematopoietic cells by the p210 BCR/ABL oncogene product of the Philadelphia chromosome. Proc. Natl Acad. Sci. USA 84, 6558–6562 (1987).
Li, S., Ilaria, R.L. Jr, Million, R.P., Daley, G.Q. & Van Etten, R.A. The p190, p210, and p230 forms of the BCR/ABL oncogene induce a similar chronic myeloid leukemia-like syndrome in mice but have different lymphoid leukemogenic activity. J. Exp. Med. 189, 1399–1412 (1999).
Daley, G.Q., Van Etten, R.A. & Baltimore, D. Blast crisis in a murine model of chronic myelogenous leukemia. Proc. Natl Acad. Sci. USA 88, 11335–11338 (1991).
Pear, W.S. et al. Efficient and rapid induction of a chronic myelogenous leukemia-like myeloproliferative disease in mice receiving p210 Bcr–Abl-transduced bone marrow. Blood 92, 3780–3792 (1998).
Zhang, X. & Ren, R. Bcr-Abl efficiently induces a myeloproliferative disease and production of excess interleukin-3 and granulocyte-macrophage colony-stimulating factor in mice: a novel model for chronic myelogenous leukemia. Blood 92, 3829–3840 (1998).
Packham, G. et al. Selective regulation of Bcl-XL by a Jak kinase-dependent pathway is bypassed in murine hematopoietic malignancies. Genes Dev. 12, 2475–2487 (1998).
Gesbert, F. & Griffin, J.D. BCR–ABL activates transcription of the Bcl-X gene through STAT5. Blood 96, 2269–2276 (2000).
Horita, M. et al. Blockade of the Bcr-Abl kinase activity induces apoptosis of chronic myelogenous leukemia cells by suppressing signal transducer and activator of transcription 5-dependent expression of Bcl-xL. J. Exp. Med. 191, 977–984 (2000).
Breitschopf, K., Haendeler, J., Malchow, P., Zeiher, A.M. & Dimmeler, S. Posttranslational modification of Bcl-2 facilitates its proteasome-dependent degradation: molecular characterization of the involved signaling pathway. Mol. Cell. Biol. 20, 1886–1896 (2000).
Cortez, D., Reuther, G. & Pendergast, A.M. The Bcr-Abl tyrosine kinase activates mitogenic signaling pathways and stimulates G1-to-S phase transition in hematopoietic cells. Oncogene 15, 2333–2342 (1997).
McGahon, A. et al. BCR-ABL maintains resistance of chronic myelogenous leukemia cells to apoptotic cell death. Blood 83, 1179–1187 (1994).
Neshat, M.S., Raitano, A.B., Wang, H.G., Reed, J.C. & Sawyers, C.L. The survival function of the Bcr-Abl oncogene is mediated by Bad-dependent and -independent pathways: roles for phosphatidylinositol 3-kinase and Raf. Mol. Cell. Biol. 20, 1179–1186 (2000).
Skorski, T. et al. Transformation of hematopoietic cells by BCR/ABL requires activation of a PI-3K/Akt-dependent pathway. EMBO J. 16, 6151–6161 (1997).
Shuai, K., Halpern, J., ten Hoeve, J., Rao, X. & Sawyers, C.L. Constitutive activation of STAT5 by the BCR-ABL oncogene in chronic myelogenous leukemia. Oncogene 13, 247–254 (1996).
Sanchez-Garcia, I. & Grutz, G. Tumorigenic activity of the BCR-ABL oncogenes is mediated by BCL2. Proc. Natl Acad. Sci. USA 92, 5287–5291 (1995).
Pugazhenthi, S. et al. Akt/protein kinase B up-regulates Bcl-2 expression through cAMP-response element-binding protein. J. Biol. Chem. 275, 10761–10766 (2000).
Muller, A.J. et al. BCR first exon sequences specifically activate the BCR/ABL tyrosine kinase oncogene of Philadelphia chromosome-positive human leukemias. Mol. Cell. Biol. 11, 1785–1792 (1991).
Zindy, F. et al. Myc signaling via the ARF tumor suppressor regulates p53-dependent apoptosis and immortalization. Genes Dev. 12, 2424–2433 (1998).
Wilson, B.E., Mochon, E. & Boxer, L.M. Induction of Bcl-2 expression by phosphorylated CREB proteins during B-cell activation and rescue from apoptosis. Mol. Cell. Biol. 16, 5546–5556 (1996).
Goga, A., McLaughlin, J., Afar, D.E., Saffran, D.C. & Witte, O.N. Alternative signals to RAS for hematopoietic transformation by the BCR-ABL oncogene. Cell 82, 981–988 (1995).
Strasser, A. et al. Enforced BCL2 expression in B-lymphoid cells prolongs antibody responses and elicits autoimmune disease. Proc. Natl Acad. Sci. USA 88, 8661–8665 (1991).
Dong, C. et al. Defective T cell differentiation in the absence of Jnk1. Science 282, 2092–2095 (1998).
Pear, W.S., Nolan, G.P., Scott, M.L. & Baltimore, D. Production of high-titer helper-free retroviruses by transient transfection. Proc. Natl Acad. Sci. USA 90, 8392–8396 (1993).
Whitlock, C.A., Robertson, D. & Witte, O.N. Murine B cell lymphopoiesis in long term culture. J. Immunol. Methods 67, 353–369 (1984).
Collins, L.S. & Dorshkind, K. A stromal cell line from myeloid long-term bone marrow cultures can support myelopoiesis and B lymphopoiesis. J. Immunol. 138, 1082–1087 (1987).
Acknowledgements
We thank M. Roussel for providing the MSCV vector, L. Boxer for the Bcl2 promoter reporter plasmid, R. Hulspas for assistance with FACS analysis, J. Wu and Q. Liu for statistical analysis, I. Joris for histological examination, Y. Liu for preparing slides, T. Barrett, J. Brackett and S. Stone for technical assistance, and K. Gemme for administrative assistance. R.J.D., C.L.S. and R.A.F. are investigators of the Howard Hughes Medical Institute. This study was supported in part by a research grant from the US National Cancer Institute and by a center grant from the US National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Hess, P., Pihan, G., Sawyers, C. et al. Survival signaling mediated by c-Jun NH2-terminal kinase in transformed B lymphoblasts. Nat Genet 32, 201–205 (2002). https://doi.org/10.1038/ng946
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng946
This article is cited by
-
Integrated molecular characterisation of the MAPK pathways in human cancers reveals pharmacologically vulnerable mutations and gene dependencies
Communications Biology (2021)
-
Combination therapy of BCR-ABL-positive B cell acute lymphoblastic leukemia by tyrosine kinase inhibitor dasatinib and c-JUN N-terminal kinase inhibition
Journal of Hematology & Oncology (2020)
-
KLF4-mediated upregulation of CD9 and CD81 suppresses hepatocellular carcinoma development via JNK signaling
Cell Death & Disease (2020)
-
c-Jun N-terminal kinase 2 promotes enterocyte survival and goblet cell differentiation in the inflamed intestine
Mucosal Immunology (2017)
-
Sensitisation of c-MYC-induced B-lymphoma cells to apoptosis by ATF2
Oncogene (2014)