Abstract
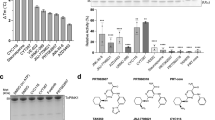
Dynamin is a large GTPase with roles in membrane fission during clathrin-mediated endocytosis, in actin dynamics and in cytokinesis. Defects in dynamin have been linked to human diseases. The synthesis of a dynamin modulator toolkit comprising two different inhibitor classes is described. The first series comprises Dynole 34-2, Dynole 2-24 and the inactive control Dynole 31-2. The Dynole compounds act on the dynamin G domain, are not GTP competitive and can be synthesized in 2–3 d. Knoevenagel condensation of 1-(3-(dimethylamino)propyl)-1H-indole-3-carbaldehyde (1) with cyanoamides (2 and 3) affords Dynole 31-2 and Dynole 34-2, respectively. Reductive amination of 1 with decylamine gives Dynole 2-24. The second series acts at an allosteric site in the G domain of dynamin and comprises Dyngo 4a and Dyngo Ø (inactive control). Both are synthesized in an overnight reaction via condensation of 3-hydroxy-2-naphthoic hydrazide with 2,4,5-trihydroxybenzaldehyde to afford Dyngo 4a, or with benzaldehyde to afford Dyngo Ø.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ferguson, S.M. & De Camilli, P. Dynamin, a membrane-remodelling GTPase. Nat. Rev. Mol. Cell Biol. 13, 75–88 (2012).
Chappie, J.S., Acharya, S., Leonard, M., Schmid, S.L. & Dyda, F. G domain dimerization controls dynamin's assembly-stimulated GTPase activity. Nature 465, 435–440 (2010).
Chappie, J.S. et al. A pseudoatomic model of the dynamin polymer identifies a hydrolysis-dependent powerstroke. Cell 147, 209–222 (2011).
Marks, B. et al. GTPase activity of dynamin and resulting conformation change are essential for endocytosis. Nature 410, 231–235 (2001).
Praefcke, G.J.K. & McMahon, H.T. The dynamin superfamily: universal membrane tubulation and fission molecules? Nat. Rev. Mol. Cell Biol. 5, 133–147 (2004).
Ferguson, K.M., Lemmon, M.A., Schlessinger, J. & Sigler, P.B. Crystal structure at 2.2 Å resolution of the pleckstrin homology domain from human dynamin. Cell 79, 199–209 (1994).
Reubold, T.F. et al. Crystal structure of the GTPase domain of rat dynamin 1. Proc. Natl. Acad. Sci. USA 102, 13093–13098 (2005).
Niemann, H.H., Knetsch, M.L.W., Scherer, A., Manstein, D.J. & Kull, F.J. Crystal structure of a dynamin GTPase domain in both nucleotide-free and GDP-bound forms. EMBO J. 20, 5813–5821 (2001).
Gao, S. et al. Structural basis of oligomerization in the stalk region of dynamin-like MxA. Nature 465, 502–506 (2010).
Ford, M.G.J., Jenni, S. & Nunnari, J. The crystal structure of dynamin. Nature 477, 561–566 (2011).
Faelber, K. et al. Crystal structure of nucleotide-free dynamin. Nature 477, 556–560 (2011).
McMahon, H.T. & Boucrot, E. Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nat. Rev. Mol. Cell Biol. 12, 517–533 (2011).
Schmid, S.L. & Frolov, V.A. Dynamin: functional design of a membrane fission catalyst. Ann. Rev. Cell Develop. Biol. 27, 79–105 (2011).
Morlot, S. et al. Membrane shape at the edge of the dynamin helix sets location and duration of the fission reaction. Cell 151, 619–629 (2012).
Smith, S.M., Renden, R. & von Gersdorff, H. Synaptic vesicle endocytosis: fast and slow modes of membrane retrieval. Trends Neurosci. 31, 559–568 (2008).
Ferguson, S.M. et al. A selective activity-dependent requirement for dynamin 1 in synaptic vesicle endocytosis. Science 316, 570–574 (2007).
Bunnage, M.E., Piatnitski Chekler, E.L. & Jones, L.H. Target validation using chemical probes. Nat. Chem. Biol. 9, 195–199 (2013).
Bain, J., McLauchlan, H., Elliott, M. & Cohen, P. The specificities of protein kinase inhibitors: an update. Biochem. J. 371, 199–204 (2003).
Harper, C.B., Popoff, M.R., McCluskey, A., Robinson, P.J. & Meunier, F.A. Targeting membrane trafficking in infection prophylaxis: dynamin inhibitors. Trends Cell Biol. 23, 90–101 (2012).
Quan, A. et al. Myristyl trimethyl ammonium bromide and octadecyl trimethyl ammonium bromide are surface-active small molecule dynamin inhibitors that block endocytosis mediated by dynamin I or dynamin II. Mol. Pharm. 72, 1425–39 (2007).
Hill, T. et al. Small molecule inhibitors of dynamin I GTPase activity: development of dimeric tyrphostins. J. Med. Chem. 48, 7781–7788 (2005).
Odell, L.R. et al. Azido and diazarinyl analogues of bis-tyrphostin as asymmetrical inhibitors of dynamin GTPase. ChemMedChem 4, 1182–1188 (2009).
Zhang, J., Lawrance, G.A., Chau, N., Robinson, P.J. & McCluskey, A. From Spanish fly to room-temperature ionic liquids (RTILs): synthesis, thermal stability and inhibition of dynamin 1 GTPase by a novel class of RTILs. New J. Chem. 32, 28–36 (2008).
Hill, T.A. et al. Iminochromene inhibitors of dynamins I and II GTPase activity and endocytosis. J. Med. Chem. 53, 4094–4102 (2010).
Odell, L.R. et al. The pthaladyns: GTP competitive inhibitors of dynamin I and II GTPase derived from virtual screening. J. Med. Chem. 53, 5267–5280 (2010).
Harper, C.B. et al. Dynamin inhibition blocks botulinum neurotoxin type A endocytosis in neurons and delays botulism. J. Biol. Chem. 286, 35966–35976 (2011).
McCluskey, A. et al. Building a better dynasore: the Dyngo compounds potently inhibit dynamin and endocytosis. Traffic 14, 1272–1289 (2013).
Robertson, M.J. et al. The rhodadyns, a new class of small molecule inhibitors of dynamin GTPase activity. Med. Chem. Lett. 3, 352–356 (2012).
McGeachie, A.B. et al. The Pyrimidyns: novel small molecule PH domain targeted pyrimidine-based dynamin inhibitors. ACS Chem. Biol. 8, 1507–1518 (2013).
Hill, T.A. et al. Inhibition of dynamin mediated endocytosis by the dynoles—synthesis and functional activity of a family of indoles. J. Med. Chem. 52, 3762–3773 (2009).
Gordon, C.P. et al. Development of second-generation indole-based dynamin GTPase inhibitors. J. Med. Chem. 56, 46–59 (2013).
Gorobets, N.Y., Yousefi, B.H., Belaj, F. & Kappe, C.O. Rapid microwave-assisted solution phase synthesis of substituted 2-pyridone libraries. Tetrahedron 60, 8633–8644 (2004).
Bhawal, B.M., Khanapure, S.P. & Biehl, E.R. Rapid, low-temperature amidation of ethyl cyano-acetate with lithium amides derived from primary and secondary amines. Syn. Commun. 20, 3235–3243 (1990).
ThalesNano. H-cube continuous-flow hydrogenation reactor (http://www.thalesnano.com/products/h-cube).
Palla, G., Predieri, G., Domiano, P., Vignali, C. & Turner, W. Conformational behaviour and E/Z isomerization of N-acyl and N-aroylhydrazones. Tetrahedron 42, 3649–3654 (1986).
Franzen, H. & Eichler, T. Replacement of hydroxyl by the hydrazine group. J. Prakt. Chem. 78, 157–164 (1909).
Sadowski, L. et al. Dynamin inhibitors impair endocytosis and mitogenic signaling of PDGF. Traffic 14, 725–736 (2013).
Bain, J. et al. The selectivity of protein kinase inhibitors: a further update. Biochem. J. 408, 297–315 (2007).
Park, R. et al. Dynamin triple knockout cells reveal off target effects of commonly used dynamin inhibitors. J. Cell Sci. 126 (part 22): 5305–5312 (2013).
Daniel, J.A., Malladi, C.S., Kettle, E., McCluskey, A. & Robinson, P.J. Analysis of synaptic vesicle endocytosis in synaptosomes by high-content screening. Nat. Prot. 7, 1439–1455 (2012).
Still, W.C., Kahn, M. & Mitra, A. Rapid chromatographic technique for preparative separations with moderate resolution. J. Org. Chem. 43, 2923–2925 (1978).
Hill, T.A. et al. Long-chain amines and long chain ammonium salts as novel inhibitors of dynamin GTPase activity. Bioorg. Med. Chem. Lett. 14, 3275–3278 (2004).
Chircop, M. et al. Inhibition of dynamin by dynole 34-2 induces cell death following cytokinesis failure in cancer cells. Mol. Can. Therap. 10, 1553–1562 (2011).
Takahashi, K. et al. . Suppression of dynamin GTPase activity by sertraline leads to inhibition of dynamin-dependent endocytosis. Biochem. Biophys. Res. Commun. 391, 382–387 (2010).
Joffre, C. et al. A direct role for Met endocytosis in tumorigenesis. Nat. Cell Biol. 13, 821–837 (2011).
Smith, C.M. & Chircop, M. Clathrin-mediated endocytic proteins are involved in regulating mitotic progression and completion. Traffic 13, 1628–1641 (2012).
Richard, J.P. et al. Intracellular curvature-generating proteins in cell-to-cell fusion. Biochemical J. 440, 185–193 (2011).
Bittner, M.A., Aikman, R.L. & Holz, R.W. A nibbling mechanism for clathrin-mediated retrieval of secretory granule membrane after exocytosis. J. Biol. Chem. 288, 9177–9188 (2013).
Kural, C. et al. Dynamics of intracellular clathrin/AP1- and clathrin/AP3-containing carriers. Cell Rep. 2, 1111–1119 (2012).
Sorkina, T., Caltagarone, J. & Sorkin, A. Flotillins regulate membrane mobility of the dopamine transporter but are not required for its protein kinase C–dependent endocytosis. Traffic 14, 709–724 (2013).
Reid, A.T. et al. Dynamin regulates specific membrane fusion events necessary for acrosomal exocytosis in mouse spermatozoa. J. Biol. Chem. 287, 37659–37672 (2012).
Acknowledgements
This work was supported by grants from the National Health and Medical Research Council (Australia), The Australia Research Council, The Australian Cancer Research Foundation, The Ramaciotti Foundation, The Children's Medical Research Institute and Newcastle Innovation, Ltd.
Author information
Authors and Affiliations
Contributions
M.J.R. and F.M.D. contributed equally to the synthesis of all analogs described in this work. P.J.R. and A.M. are responsible for the concept, design and use of the dynamin inhibitors reported herein.
Corresponding authors
Ethics declarations
Competing interests
We have a commercial agreement with Abcam Biochemicals (Bristol, UK) for the supply of our dynamin inhibitors. This includes many of the compounds listed in this paper. Quinodyn, Dynole, Rhodadyn, MiTMAB, Bis-T, Dyngo and Iminodyn are trademarks of Children's Medical Research Institute and Newcastle Innovation, Ltd. Most of the lead dynamin inhibitors described in this paper are available from Abcam Biochemicals (Bristol, UK).
Rights and permissions
About this article
Cite this article
Robertson, M., Deane, F., Robinson, P. et al. Synthesis of Dynole 34-2, Dynole 2-24 and Dyngo 4a for investigating dynamin GTPase. Nat Protoc 9, 851–870 (2014). https://doi.org/10.1038/nprot.2014.046
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2014.046
This article is cited by
-
Small molecule inhibition of Dynamin-dependent endocytosis targets multiple niche signals and impairs leukemia stem cells
Nature Communications (2020)
-
A pH-responsive nanoparticle targets the neurokinin 1 receptor in endosomes to prevent chronic pain
Nature Nanotechnology (2019)
-
Dynasore impairs VEGFR2 signalling in an endocytosis-independent manner
Scientific Reports (2017)
-
Dynamin 2 is essential for mammalian spermatogenesis
Scientific Reports (2016)
-
Dynamin II is required for 17β-estradiol signaling and autophagy-based ERα degradation
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.