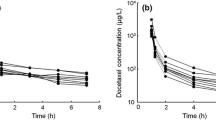
Abstract
Docetaxel belongs to the class of taxane antineoplastic agents that act by inducing microtubular stability and disrupting the dynamics of the microtubular network. The drug has shown a broad spectrum of antitumour activity in preclinical models as well as clinically, with responses observed in various disease types, including advanced breast cancer and non-small cell lung cancer. The pharmacokinetics and metabolism of docetaxel are extremely complex and have been the subject of intensive investigation in recent years. Docetaxel is subject to extensive metabolic conversion by the cytochrome P450 (CYP) 3A isoenzymes, which results in several pharmacologically inactive oxidation products. Elimination routes of docetaxel are also dependent on the presence of drug-transporting proteins, notably P-glycoprotein, present on the bile canalicular membrane. The various processes mediating drug elimination, either through metabolic breakdown or excretion, impact substantially on interindividual variability in drug handling. Strategies to individualise docetaxel administration schedules based on phenotypic or genotype-dependent differences in CYP3A expression are underway and may ultimately lead to more selective chemotherapeutic use of this agent.





Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and does not imply endorsement.
References
Nabholtz JM, Senn HJ, Bezwoda WR, et al. Prospective randomized trial of docetaxel versus mitomycin plus vinblastine in patients with metastatic breast cancer progressing despite previous anthracycline-containing chemotherapy. 304 Study Group. J Clin Oncol 1999; 17: 1413–24
Fossella FV, DeVore R, Kerr RN, et al. Randomized phase III trial of docetaxel versus vinorelbine or ifosfamide in patients with advanced non-small-cell lung cancer previously treated with platinum-containing chemotherapy regimens. The TAX 320 Non-Small Cell Lung Cancer Study Group. J Clin Oncol 2000; 18: 2354–62
Ando M, Watanabe T, Nagata K, et al. Efficacy of docetaxel 60 mg/m2 in patients with metastatic breast cancer according to the status of anthracycline resistance. J Clin Oncol 2001; 19: 336–42
Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004; 351: 1502–12
Engels FK, Verweij J. Docetaxel administration schedule: from fever to tears? A review of randomised studies. Eur J Cancer 2005; 41: 1117–26
Clarke SJ, Rivory LP. Clinical pharmacokinetics of docetaxel. Clin Pharmacokinet 1999; 36: 99–114
Vergniol JC, Bruno R, Montay G, et al. Determination of Taxotere in human plasma by a semi-automated high-performance liquid chromatographic method. J Chromatogr 1992; 582: 273–8
Hou W, Watters JW, McLeod HL. Simple and rapid docetaxel assay in plasma by protein precipitation and high-performance liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2004; 804: 263–7
Wang LZ, Goh BC, Grigg ME, et al. A rapid and sensitive liquid chromatography/tandem mass spectrometry method for determination of docetaxel in human plasma. Rapid Commun Mass Spectrom 2003; 17: 1548–52
Gustafson DL, Long ME, Zirrolli JA, et al. Analysis of docetaxel pharmacokinetics in humans with the inclusion of later sampling time-points afforded by the use of a sensitive tandem LCMS assay. Cancer Chemother Pharmacol 2003; 52: 159–66
Ciccolini J, Catalin J, Blachon MF, et al. Rapid high-performance liquid chromatographic determination of docetaxel (Taxotere) in plasma using liquid-liquid extraction. J Chromatogr B Biomed Sci Appl 2001; 759: 299–306
Loos WJ, Verweij J, Nooter K, et al. Sensitive determination of docetaxel in human plasma by liquid-liquid extraction and reversed-phase high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 1997; 693: 437–41
Baker SD, Zhao M, He P, et al. Simultaneous analysis of docetaxel and the formulation vehicle polysorbate 80 in human plasma by liquid chromatography/tandem mass spectrometry. Anal Biochem 2004; 324: 276–84
Mortier KA, Renard V, Verstraete AG, et al. Development and validation of a liquid chromatography-tandem mass spectrometry assay for the quantification of docetaxel and paclitaxel in human plasma and oral fluid. Anal Chem 2005; 77: 4677–83
Kuppens IE, van Maanen MJ, Rosing H, et al. Quantitative analysis of docetaxel in human plasma using liquid chromatography coupled with tandem mass spectrometry. Biomed Chromatogr 2005; 19: 355–61
Parise RA, Ramanathan RK, Zamboni WC, et al. Sensitive liquid chromatography-mass spectrometry assay for quantitation of docetaxel and paclitaxel in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2003; 783: 231–6
Garg MB, Ackland SP. Simple and sensitive high-performance liquid chromatography method for the determination of docetaxel in human plasma or urine. J Chromatogr B Biomed Sci Appl 2000; 748: 383–8
Rosing H, Lustig V, Koopman FP, et al. Bio-analysis of docetaxel and hydroxylated metabolites in human plasma by high-performance liquid chromatography and automated solid-phase extraction. J Chromatogr B Biomed Sci Appl 1997; 696: 89–98
Ardiet CJ, Tranchand B, Zanetta S, et al. A sensitive docetaxel assay in plasma by solid-phase extraction and high performance liquid chromatography-UV detection: validation and suitability in phase I clinical trial pharmacokinetics. Invest New Drugs 1999; 17: 325–33
Rouini MR, Lotfolahi A, Stewart DJ, et al. A rapid reversed phase high performance liquid chromatographic method for the determination of docetaxel (Taxotere) in human plasma using a column switching technique. J Pharm Biomed Anal 1998; 17: 1243–7
Urien S, Barre J, Morin C, et al. Docetaxel serum protein binding with high affinity to alpha 1-acid glycoprotein. Invest New Drugs 1996; 14: 147–51
Acharya MR, Baker SD, Verweij J, et al. Determination of fraction unbound docetaxel using micro-equilibrium dialysis. Anal Biochem 2004; 331: 192–4
Loos WJ, Baker SD, Verweij J, et al. Clinical pharmacokinetics of unbound docetaxel: role of polysorbate 80 and serum proteins. Clin Pharmacol Ther 2003; 74: 364–71
Kremer JM, Wilting J, Janssen LH. Drug binding to human alpha-1-acid glycoprotein in health and disease. Pharmacol Rev 1988; 40: 1–47
Baker SD, Van Schaik RH, Rivory LP, et al. Factors affecting cytochrome P-450 3A activity in cancer patients. Clin Cancer Res 2004; 10: 8341–50
Bruno R, Vivler N, Vergniol JC, et al. A population pharmacokinetic model for docetaxel (Taxotere): model building and validation. J Pharmacokinet Biopharm 1996; 24: 153–72
Bruno R, Hille D, Riva A, et al. Population pharmacokinetics/ pharmacodynamics of docetaxel in phase II studies in patients with cancer. J Clin Oncol 1998; 16: 187–96
Bruno R, Olivares R, Berille J, et al. Alpha-1-acid glycoprotein as an independent predictor for treatment effects and a prognostic factor of survival in patients with non-small cell lung cancer treated with docetaxel. Clin Cancer Res 2003; 9: 1077–82
Rivory LP, Slaviero KA, Clarke SJ. Hepatic cytochrome P450 3A drug metabolism is reduced in cancer patients who have an acute-phase response. Br J Cancer 2002; 87: 277–80
Slaviero KA, Clarke SJ, Rivory LP. Inflammatory response: an unrecognised source of variability in the pharmacokinetics and pharmacodynamics of cancer chemotherapy. Lancet Oncol 2003; 4: 224–32
Baker SD, Li J, Ten Tije AJ, et al. Relation of systemic exposure to unbound docetaxel and neutropenia. Clin Pharmacol Ther 2005; 77: 43–53
Ten Tije AJ, Loos WJ, Verweij J, et al. Disposition of polyoxyethylated excipients in humans: implications for drug safety and formulation approaches. Clin Pharmacol Ther 2003; 74: 509–10
Bravo Gonzalez RC, Huwyler J, Boess F, et al. In vitro investigation on the impact of the surface-active excipients Cremophor EL, Tween 80 and SOlutol HS 15 on the metabolism of midazolam. Biopharm Drug Dispos 2004; 25: 37–49
Wandel C, Kim RB, Stein CM. “Inactive” excipients such as Cremophor can affect in vivo drug disposition. Clin Pharmacol Ther 2003; 73: 394–6
Hugger ED, Novak BL, Burton PS, et al. A comparison of commonly used polyethoxylated pharmaceutical excipients on their ability to inhibitor P-glycoprotein activity in vitro. J Pharm Sci 2002; 91: 1991–2002
Shou M, Martinet M, Korzekwa KR, et al. Role of human cytochrome P450 3A4 and 3A5 in the metabolism of taxotere and its derivatives: enzyme specificity, interindividual distribution and metabolic contribution in human liver. Pharmacogenetics 1998; 8: 391–401
Sparreboom A, Van Tellingen O, Scherrenburg EJ, et al. Isolation, purification and biological activity of major docetaxel metabolites from human feces. Drug Metab Dispos 1996; 24: 655–8
Xie HG, Wood AJ, Kim RB, et al. Genetic variability in CYP3A5 and its possible consequences. Pharmacogenomics 2004; 5: 243–72
Lamba JK, Lin YS, Schuetz EG, et al. Genetic contribution to variable human CYP3A-mediated metabolism. Adv Drug Deliv Rev 2002; 54: 1271–94
Amirimani B, Walker AH, Weber BL, et al. RESPONSE: re modification of clinical presentation of prostate tumors by a novel genetic variant in CYP3A4. J Natl Cancer Inst 1999; 91: 1588–90
Goh BC, Lee SC, Wang LZ, et al. Explaining interindividual variability of docetaxel pharmacokinetics and pharmacodynamics in Asians through phenotyping and genotyping strategies. J Clin Oncol 2002; 20: 3683–90
Floyd MD, Gervasini G, Masica AL, et al. Genotype-phenotype associations for common CYP3A4 and CYP3A5 variants in the basal and induced metabolism of midazolam in European-and African-American men and women. Pharmacogenetics 2003; 13: 595–606
Hustert E, Haberl M, Burk O, et al. The genetic determinants of the CYP3A5 polymorphism. Pharmacogenetics 2001; 11: 773–9
Kuehl P, Zhang J, Lin Y, et al. Sequence diversity in CYP3A promoters and characterization of the genetic basis of polymorphic CYP3A5 expression. Nat Genet 2001; 27: 383–91
Puisset F, Chatelut E, Dalenc F, et al. Dexamethasone as a probe for docetaxel clearance. Cancer Chemother Pharmacol 2004; 54: 265–72
Wong M, Balleine RL, Collins M, et al. CYP3A5 genotype and midazolam clearance in Australian patients receiving chemotherapy. Clin Pharmacol Ther 2004; 75: 529–38
Streetman DS, Bertino Jr JS, Nafziger AN. Phenotyping of drug-metabolizing enzymes in adults: a review of in-vivo cytochrome P450 phenotyping probes. Pharmacogenetics 2000; 10: 187–216
Shih PS, Huang JD. Pharmacokinetics of midazolam and 1′-hydroxymidazolam in Chinese with different CYP3A5 genotypes. Drug Metab Dispos 2002; 30: 1491–6
Eap CB, Buclin T, Hustert E, et al. Pharmacokinetics of midazolam in CYP3A4- and CYP3A5-genotyped subjects. Eur J Clin Pharmacol 2004; 60_(4): 231–6
Ringel I, Horwitz SB. Studies with RP 56976 (taxotere): a semisynthetic analogue of taxol. J Natl Cancer Inst 1991; 83: 288–91
Marzolini C, Paus E, Buclin T, et al. Polymorphisms in human MDR1 (P-glycoprotein): recent advances and clinical relevance. Clin Pharmacol Ther 2004; 75: 13–33
Bardelmeijer HA, Ouwehand M, Buckle T, et al. Low systemic exposure of oral docetaxel in mice resulting from extensive first-pass metabolism is boosted by ritonavir. Cancer Res 2002; 62: 6158–64
Sparreboom A, van Asperen J, Mayer U, et al. Limited oral bioavailability and active epithelial excretion of paclitaxel (Taxol) caused by P-glycoprotein in the intestine. Proc Natl Acad Sci U S A 1997; 94: 2031–5
van Zuylen L, Verweij J, Nooter K, et al. Role of intestinal P-glycoprotein in the plasma and fecal disposition of docetaxel in humans. Clin Cancer Res 2000; 6: 2598–603
Huisman MT, Chhatta AA, van Tellingen O, et al. MRP2 (ABCC2) transports taxanes and confers paclitaxel resistance and both processes are stimulated by probenecid. Int J Cancer 2005 Sep 20; 116(5): 824–9
Hopper-Borge E, Chen ZS, Shchaveleva I, et al. Analysis of the drug resistance profile of multidrug resistance protein 7 (ABCC10): resistance to docetaxel. Cancer Res 2004; 64: 4927–30
Hirth J, Watkins PB, Strawderman M, et al. The effect of an individual’s cytochrome CYP3A4 activity on docetaxel clearance. Clin Cancer Res 2000; 6: 1255–8
Wagner D. CYP3A4 and the erythromycin breath test. Clin Pharmacol Ther 1998; 64: 129–30
Yamamoto N, Tamura T, Kamiya Y, et al. Correlation between docetaxel clearance and estimated cytochrome P450 activity by urinary metabolite of exogenous cortisol. J Clin Oncol 2000; 18: 2301–8
Rudek MA, Sparreboom A, Garrett-Mayer ES, et al. Factors affecting pharmacokinetic variability following doxorubicin and docetaxel-based therapy. Eur J Cancer 2004; 40: 1170–8
Ten Tije AJ, Verweij J, Carducci MA, et al. Prospective evaluation of the pharmacokinetics and toxicity profile of docetaxel in the elderly. J Clin Oncol 2005; 23: 1070–7
Slaviero KA, Clarke SJ, McLachlan AJ, et al. Population pharmacokinetics of weekly docetaxel in patients with advanced cancer. Br J Clin Pharmacol 2004; 57: 44–53
Yamamoto N, Tamura T, Murakami H, et al. Randomized pharmacokinetic and pharmacodynamic study of docetaxel: dosing based on body-surface area compared with individualized dosing based on cytochrome P450 activity estimated using a urinary metabolite of exogenous cortisol. J Clin Oncol 2005; 23: 1061–9
Kuroi K, Bando H, Saji S, et al. Weekly schedule of docetaxel in breast cancer: evaluation of response and toxicity. Breast Cancer 2003; 10: 10–4
Zimatore M, Danova M, Vassallo E, et al. Weekly taxanes in metastatic breast cancer (review). Oncol Rep 2002; 9: 1047–52
Climent MA, Tabernero J, Albanell J, et al. Preliminary results of a phase II randomized trial of docetaxel (taxotere) as a single agent chemotherapy (CT) administered weekly or 3-weekly in patients (pts) with metastatic breast cancer (MBC) [abstract]. Proc Am Soc Clin Oncol 2002; 21: 52A
Gervais R, Ducolone AM, Breton J-L, et al. Multicenter, randomised, phase II trial of docetaxel (Taxotere) 75 mg/m2 q3w versus 40 mg/m2 weekly in patients with pretreated non small cell lung cancer (NSCLC) [abstract]. Proc Am Soc Clin Oncol 2002; 21: 10A
Baker SD, Zhao M, Lee CK, et al. Comparative pharmacokinetics of weekly and every-three-weeks docetaxel. Clin Cancer Res 2004; 10: 1976–83
Minami H, Ohe Y, Niho S, et al. Comparison of pharmacokinetics and pharmacodynamics of docetaxel and cisplatin in elderly and non-elderly patients: why is toxicity increased in elderly patients?. J Clin Oncol 2004; 22: 2901–8
Hainsworth JD, Burris III HA, Litchy S, et al. Weekly docetaxel in the treatment of elderly patients with advanced non small cell lung carcinoma. A Minnie Pearl Cancer Research Network Phase II Trial. Cancer 2000; 89: 328–33
Hainsworth JD, Burris III HA, Yardley DA, et al. Weekly docetaxel in the treatment of elderly patients with advanced breast cancer: a Minnie Pearl Cancer Research Network phase II trial. J Clin Oncol 2001; 19: 3500–5
Hainsworth JD, Erland JB, Barton JH, et al. Combination treatment with weekly docetaxel and gemcitabine for advanced non-small-cell lung cancer in elderly patients and patients with poor performance status: results of a Minnie Pearl Cancer Research Network phase II trial. Clin Lung Cancer 2003; 5: 33–8
Wildiers H, Paridaens R. Taxanes in elderly breast cancer patients. Cancer Treat Rev 2004; 30: 333–42
Baker SD, Ravdin P, Aylesworth C, et al. A phase I and pharmacokinetic study of docetaxel in cancer patients with liver dysfunction due to malignancies [abstract]. Proc Am Soc Clin Oncol 1998; 17: 192A
Synold TW, Newman E, Lenz H-J, et al. Prospective evaluation of docetaxel (D) pharmacokinetics (PK) and toxicity in patients with tumor-related hepatic dysfunction (HD) [abstract]. Proc Am Soc Clin Oncol 1999; 18: 168A
Bissery MC, Nohynek G, Sanderink GJ, et al. Docetaxel (Taxotere): a review of preclinical and clinical experience (Pt I): preclinical experience. Anticancer Drugs 1995; 6: 339-55, 363–68
Liu G, Franssen E, Fitch MI, et al. Patient preferences for oral versus intravenous palliative chemotherapy. J Clin Oncol 1997; 15: 110–5
Van Asperen J, Van Tellingen O, Beijnen JH. The pharmacological role of P-glycoprotein in the intestinal epithelium. Pharmacol Res 1998; 37: 429–35
Kruijtzer CM, Beijnen JH, Schellens JH. Improvement of oral drug treatment by temporary inhibition of drug transporters and/or cytochrome P450 in the gastrointestinal tract and liver: an overview. Oncologist 2002; 7: 516–30
Malingre MM, Richel DJ, Beijnen JH, et al. Coadministration of cyclosporine strongly enhances the oral bioavailability of docetaxel. J Clin Oncol 2001; 19: 1160–6
Malingre MM, Beijnen JH, Schellens JH. Oral delivery of taxanes. Invest New Drugs 2001; 19: 155–62
Kuppens IE, Bosch TM, van Maanen MJ, et al. Oral bioavailability of docetaxel in combination with OC144-093 (ONT093). Cancer Chemother Pharmacol 2005; 55: 72–8
Sampath D, Discafani CM, Loganzo F, et al. MAC-321, a novel taxane with greater efficacy than paclitaxel and docetaxel in vitro and in vivo. Mol Cancer Ther 2003; 2: 873–84
Iimura S, Uoto K, Ohsuki S, et al. Orally active docetaxel analogue: synthesis of 10-deoxy-10-C-morpholinoethyl docetaxel analogues. Bioorg Med Chem Lett 2001; 11: 407–10
Petrangolini G, Cassinelli G, Pratesi G, et al. Antitumour and antiangiogenic effects of IDN 5390, a novel C-seco taxane, in a paclitaxel-resistant human ovarian tumour xenograft. Br J Cancer 2004; 90: 1464–8
Vredenburg MR, Ojima I, Veith J, et al. Effects of orally active taxanes on P-glycoprotein modulation and colon and breast carcinoma drug resistance. J Natl Cancer Inst 2001; 93: 1234–45
Mastalerz H, Cook D, Fairchild CR, et al. The discovery of BMS-275183: an orally efficacious novel taxane. Bioorg Med Chem 2003; 11: 4315–23
Lee FY, Borzilleri R, Fairchild CR, et al. BMS-247550: a novel epothilone analog with a mode of action similar to paclitaxel but possessing superior antitumor efficacy. Clin Cancer Res 2001; 7: 1429–37
Maenpaa JU. Docetaxel: promising and novel combinations in ovarian cancer. Br J Cancer 2003; 89 Suppl. 3: S29–34
Mohamed F, Stuart OA, Sugarbaker PH. Pharmacokinetics and tissue distribution of intraperitoneal docetaxel with different carrier solutions. J Surg Res 2003; 113: 114–20
Mohamed F, Sugarbaker PH. Intraperitoneal taxanes. Surg Oncol Clin N Am 2003; 12: 825–33
Marchettini P, Stuart OA, Mohamed F, et al. Docetaxel: pharmacokinetics and tissue levels after intraperitoneal and intravenous administration in a rat model. Cancer Chemother Pharmacol 2002; 49: 499–503
de Bree E, Rosing H, Beijnen JH, et al. Pharmacokinetic study of docetaxel in intraoperative hyperthermic i.p. chemotherapy for ovarian cancer. Anticancer Drugs 2003; 14: 103–10
de Bree E, Romanos J, Michalakis J, et al. Intraoperative hyperthermic intraperitoneal chemotherapy with docetaxel as second-line treatment for peritoneal carcinomatosis of gynaecological origin. Anticancer Res 2003; 23: 3019–27
Morgan Jr RJ, Doroshow JH, Synold T, et al. Phase I trial of intraperitoneal docetaxel in the treatment of advanced malignancies primarily confined to the peritoneal cavity: dose-limiting toxicity and pharmacokinetics. Clin Cancer Res 2003; 9: 5896–901
Gelderblom H, Verweij J, van Zomeren DM, et al. Influence of Cremophor El on the bioavailability of intraperitoneal paclitaxel. Clin Cancer Res 2002; 8: 1237–41
Van Den Neste E, de Valeriola D, Kerger J, et al. A phase I and pharmacokinetic study of docetaxel administered in combination with continuous intravenous infusion of 5-fluorouracil in patients with advanced solid tumors. Clin Cancer Res 2000; 6: 64–71
Millward MJ, Zalcberg J, Bishop JF, et al. Phase I trial of docetaxel and cisplatin in previously untreated patients with advanced non-small-cell lung cancer. J Clin Oncol 1997; 15: 750–8
Pronk LC, Stoter G, Verweij J. Docetaxel (Taxotere): single agent activity, development of combination treatment and reducing side-effects. Cancer Treat Rev 1995; 21: 463–78
Oka M, Fukuda M, Nagashima S, et al. Phase I study of second-line chemotherapy with docetaxel and carboplatin in advanced non-small-cell lung cancer. Cancer Chemother Pharmacol 2001; 48: 446–50
Ando M, Saka H, Ando Y, et al. Sequence effect of docetaxel and carboplatin on toxicity, tumor response and pharmacokinetics in non-small-cell lung cancer patients: a phase I study of two sequences. Cancer Chemother Pharmacol 2005; 55: 552–8
Esposito M, Venturini M, Vannozzi MO, et al. Comparative effects of paclitaxel and docetaxel on the metabolism and pharmacokinetics of epirubicin in breast cancer patients. J Clin Oncol 1999; 17: 1132
Lunardi G, Venturini M, Vannozzi MO, et al. Influence of alternate sequences of epirubicin and docetaxel on the pharmacokinetic behaviour of both drugs in advanced breast cancer. Ann Oncol 2002; 13: 280–5
Airoldi M, Cattel L, Marchionatti S, et al. Docetaxel and vinorelbine in recurrent head and neck cancer: pharmacokinetic and clinical results. Am J Clin Oncol 2003; 26: 378–81
Dumez H, Louwerens M, Pawinsky A, et al. The impact of drug administration sequence and pharmacokinetic interaction in a phase I study of the combination of docetaxel and gemcitabine in patients with advanced solid tumors. Anticancer Drugs 2002; 13: 583–93
Louwerens M, Smorenburg C, Sparreboom A, et al. Phase I pharmacokinetic and sequence finding study of the combination of docetaxel and methotrexate in patients with solid tumours. Eur J Cancer 2002; 38: 497–504
Lortholary A, Maillard P, Delva R, et al. Docetaxel in combination with 5-fluorouracil in patients with metastatic breast cancer previously treated with anthracycline-based chemotherapy: a phase I, dose-finding study. Eur J Cancer 2000; 36: 1773–80
Pronk LC, Vasey P, Sparreboom A, et al. A phase I and pharmacokinetic study of the combination of capecitabine and docetaxel in patients with advanced solid tumours. Br J Cancer 2000; 83: 22–9
Ramanathan RK, Ramalingam S, Egorin MJ, et al. Phase I study of weekly (day 1 and 8) docetaxel in combination with capecitabine in patients with advanced solid malignancies. Cancer Chemother Pharmacol 2005; 55: 354–60
Schrijvers D, Pronk L, Highley M, et al. Pharmacokinetics of ifosfamide are changed by combination with docetaxel: results of a phase I pharmacologic study. Am J Clin Oncol 2000; 23: 358–63
Petrylak DP, Macarthur RB, O’Connor J, et al. Phase I trial of docetaxel with estramustine in androgen-independent prostate cancer. J Clin Oncol 1999; 17: 958–67
Adjei AA, Klein CE, Kastrissios H, et al. Phase I and pharmacokinetic study of irinotecan and docetaxel in patients with advanced solid tumors: preliminary evidence of clinical activity. J Clin Oncol 2000; 18: 1116–23
Garcia AA, Pujari M, Jeffers S, et al. Phase I clinical and pharmacokinetic trial of docetaxel and irinotecan administered on a weekly schedule. Cancer Chemother Pharmacol 2005; 56: 75–82
Yoshida K, Hirabayashi N, Takiyama W, et al. Phase I study of combination therapy with S-1 and docetaxel (TXT) for advanced or recurrent gastric cancer. Anticancer Res 2004; 24: 1843–51
Tan AR, Yang X, Berman A, et al. Phase I trial of the cyclindependent kinase inhibitor flavopiridol in combination with docetaxel in patients with metastatic breast cancer. Clin Cancer Res 2004; 10: 5038–47
Tolcher AW, Kuhn J, Schwartz G, et al. A phase I pharmacokinetic and biological correlative study of oblimersen sodium (genasense, G3139), an antisense oligonucleotide to the BCL2 mRNA, and of docetaxel in patients with hormone-refractory prostate cancer. Clin Cancer Res 2004; 10: 5048–57
Schwartz GH, Jones CB, Garrison M, et al. A phase I and pharmacokinetic study of the nonpolyglutamatable thymidylate synthase inhibitor ZD9331 plus docetaxel in patients with advanced solid malignancies. Invest New Drugs 2004; 22: 437–48
Cresta S, Grasselli G, Mansutti M, et al. A randomized phase II study of combination, alternating and sequential regimens of doxorubicin and docetaxel as first-line chemotherapy for women with metastatic breast cancer. Ann Oncol 2004; 15: 433–9
Itoh K, Sasaki Y, Fujii H, et al. Study of dose escalation and sequence switching of administration of the combination of docetaxel and doxorubicin in advanced breast cancer. Clin Cancer Res 2000; 6: 4082–90
Pavlick AC, Chodkiewicz C, Liebes L, et al. A phase I and pharmacokinetic study of docetaxel combined with Doxil (pegylated liposomal doxorubicin) without and with granulocyte colony stimulating factor. Anticancer Drugs 2004; 15: 119–25
Briasoulis E, Karavasilis V, Tzamakou E, et al. Interaction pharmacokinetics of pegylated liposomal doxorubicin (Caelyx) on coadministration with paclitaxel or docetaxel. Cancer Chemother Pharmacol 2004; 53: 452–7
Zamboni WC, Egorin MJ, Van Echo DA, et al. Pharmacokinetic and pharmacodynamic study of the combination of docetaxel and topotecan in patients with solid tumors. J Clin Oncol 2000; 18: 3288–94
Tkaczuk KH, Zamboni WC, Tait NS, et al. Phase I study of docetaxel and topotecan in patients with solid tumors. Cancer Chemother Pharmacol 2000; 46: 442–8
Posey JA, Wang H, Hamilton J, et al. Phase-I dose escalation and sequencing study of docetaxel and continuous infusion topotecan in patients with advanced malignancies. Cancer Chemother Pharmacol 2005; 56: 182–8
Engels FK, Ten Tije AJ, Baker SD, et al. Effect of cytochrome P450 3A4 inhibition on the pharmacokinetics of docetaxel. Clin Pharmacol Ther 2004; 75: 448–54
Nygren P, Hande K, Petty KJ, et al. Lack of effect of aprepitant on the pharmacokinetics of docetaxel in cancer patients. Cancer Chemother Pharmacol 2005; 55: 609–16
Drug interactions: defining genetic influences on pharmacologic responses [online]. Available from URL: http://medicine.iupui.edu/flockhart [Accessed 2006 Jan 24]
van Zuylen L, Sparreboom A, van der Gaast A, et al. The orally administered P-glycoprotein inhibitor R101933 does not alter the plasma pharmacokinetics of docetaxel. Clin Cancer Res 2000; 6: 1365–71
van Zuylen L, Sparreboom A, van der Gaast A, et al. Disposition of docetaxel in the presence of P-glycoprotein inhibition by intravenous administration of R101933. Eur J Cancer 2002; 38: 1090–9
Fracasso PM, Goldstein LJ, de Alwis DP, et al. Phase I study of docetaxel in combination with the P-glycoprotein inhibitor, zosuquidar, in resistant malignancies. Clin Cancer Res 2004; 10: 7220–8
Kemper EM, Verheij M, Boogerd W, et al. Improved penetration of docetaxel into the brain by co-administration of inhibitors of P-glycoprotein. Eur J Cancer 2004; 40: 1269–74
Sandstrom M, Lindman H, Nygren P, et al. Model describing the relationship between pharmacokinetics and hematologic toxicity of the epirubicin-docetaxel regimen in breast cancer patients. J Clin Oncol 2005; 23: 413–21
Veyrat-Follet C, Bruno R, Olivares R, et al. Clinical trial simulation of docetaxel in patients with cancer as a tool for dosage optimization. Clin Pharmacol Ther 2000; 68: 677–87
Vogel CL, Wojtukiewicz MZ, Carroll RR, et al. First and subsequent cycle use of pegfilgrastim prevents febrile neutropenia in patients with breast cancer: a multicenter, doubleblind, placebo-controlled phase III study. J Clin Oncol 2005; 23: 1178–84
Ozer H, Armitage JO, Bennett CL, et al. 2000 update of recommendations for the use of hematopoietic colony-stimulating factors: evidence-based, clinical practice guidelines. American Society of Clinical Oncology Growth Factors Expert Panel. J Clin Oncol 2000; 18: 3558–85
Acknowledgements
Dr Baker receives research funding from Sanofi Aventis and the terms of this arrangement are being managed by the Johns Hopkins University in accordance with its conflict of interest policies. All other authors do not have any conflicts of interest directly relevant to the contents of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baker, S.D., Sparreboom, A. & Verweij, J. Clinical Pharmacokinetics of Docetaxel. Clin Pharmacokinet 45, 235–252 (2006). https://doi.org/10.2165/00003088-200645030-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200645030-00002