Abstract
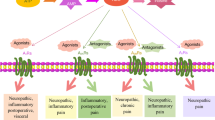
Substantial evidence exists for the physiological role of adenosine in the modulation of primary afferent transmission. Since the first description of the anti-nociceptive effects of adenosine, there has been considerable interest in the development of adenosine analogues as potential analgesics for the treatment of various pain states. The direction of effect of adenosine in the periphery is complicated by the existence of multiple receptors and species differences. The analgesic actions of agents acting on adenosine receptor systems are largely attributed to actions at the spinal cord. Two subtypes of adenosine receptors (A1 and A2) have been identified in the substantia gelatinosa of the spinal cord where they were shown to be localised primarily on intrinsic neurons. Although evidence exists for the involvement of A2 receptors in spinally mediated antinociception, it appears to be predominantly the A1 receptor subtype which plays a major role in inhibiting the nociceptive input in the dorsal spinal cord.
The antinociceptive properties of adenosine and receptor-selective analogues have been demonstrated across a wide range of animal models, including acute nociceptive tests and in models of inflammation and neuropathy. These results, observed across several models of pain, strongly support the potential clinical use of these agents in various pain states. In humans, systemic or intrathecal administration of adenosine was shown to be effective against experimentally induced pain in healthy volunteers. Subsequently, there is evidence for the effectiveness of systemic and spinally administered adenosine in patients with neuropathic pain.
A number of studies have demonstrated possible interactions between adenosine and glutamate in the spinal cord, and between N-methyl-D-aspartate receptor activation and adenosine release elsewhere in the brain. There is also some evidence that drugs acting to inhibit the metabolism of adenosine may have therapeutic potential in pain states. In this context, the release of adenosine appears to be elevated in hyperexcitable neuronal systems. This would allow therapies to selectively target active neurons in pain systems and therefore would be expected to have low adverse effect liability.
Similar content being viewed by others
References
Dickenson AH. Mechanisms of the analgesic actions of opiates and opioids. Br Med Bull 1991; 47: 690–702
Dickenson AH. Where and how do opioids act? In: Gebhart GF, Hammond DL, Jenson TS, editors. Proceedings of the 7th World Congress on Pain. Progress in Pain Research and Management. Vol. 2. Seattle (WA): IASP Press, 1994: 525–52
Yaksh TL. The spinal actions of opioids. In: Herz A, editor. Handbook of experimental pharmacology. Opioids. Vol. 104/11. Heidelberg: Springer-Verlag, 1993: 53–90
Yaksh TL, Rudy TA. Studies on the direct spinal action of narcotics in the production of analgesia in the rat. J Pharmacol Exp Ther 1977; 202: 411–28
Fredholm BB, Abbracchio MP, Burnstock G, et al. Nomenclature and classification of purinoreceptors. Pharmacol Rev 1994; 46(2): 143–56
Ralevic V, Burnstock G. Receptors for purines and pyramidines. Pharmacol Rev 1998; 50: 413–92
Choca JI, Green RD, Proudfit HK. Adenosine A1 and A2 receptors of the substantia gelatinosa are located predominantly on intrinsic neurons: an autoradiography study. J Pharmacol Exp Ther 1988; 247: 757–64
Fastbom J, Pazos A, Probst A, et al. Adenosine A1 receptors in the human brain: a quantitative autoradiographic study. Neuroscience 1987; 22: 827–39
Goodman RR, Kuhar MJ, Hester L, et al. Adenosine receptors: autoradiographic evidence for their location on axon terminals of excitatory neurones. Science 1983; 220: 967–9
Goodman RR, Snyder SH. Autoradiographic localization of adenosine receptors in rat brain using [3H]cyclohexyladenosine. J Neurosci 1982; 2: 1230–41
Geiger JD, LaBella FS, Nagy JI. Characterization and localization of adenosine receptors in rat spinal cord. J Neurosci 1984; 4: 2303–10
Linden J. Cloned adenosine A3 receptors: pharmacological properties, species differences and receptor functions. Trends Pharmacol Sci 1994; 15: 298–306
Murphy S, Pearce B. Functional receptors for neurotransmitters on astroglial cells. Neuroscience 1987; 22: 381–94
Sweeney MI, White TD, Sawynok J. Morphine-evoked release of adenosine from the spinal cord occurs via a nucleoside carrier with differential sensitivity to dipyridamole and nitro-benzylthioinosine. Brain Res 1993; 614: 301–7
Braas KM, Newby AC, Wilson VS, et al. Adenosine-containing neurones in the brain localized by immunocytochemistry. J Neurosci 1986; 6: 1952–61
Garthwaite J, Garthwaite G, Palmer RMJ, et al. NMDA receptor activation induces nitric oxide synthesis from arginine in rat brain slices. Eur J Pharmacol 1989; 172: 413–6
Santicioli P, Del Bianco E, Maggi CA. Adenosine A1 receptors mediate the presynaptic inhibition of calcitonin gene related peptide release by adenosine in the rat spinal cord. Eur J Pharm 1993; 231: 139–42
Geiger JD, Nagy JI. Distribution of adenosine deaminase activity in rat brain and spinal cord. J Neurosci 1986; 6: 2707–14
Nagy JI, Daddona PE. Anatomical and cytochemical relationships of adenosine deaminase-containing primary afferent neurons in the rat. Neurosci Int 1985; 15: 799–813
Nagy JI, Buss M, LaBella LA, et al. Immunohistochemical localization of adenosine deaminase in primary afferent neurons of the rat. Neurosci Lett 1984; 48: 133–8
Geiger JD, Nagy JI. Heterogeneous distribution of adenosine transport sites labelled by [3H]nitrobenzylthioinosine in rat brain: an autoradiographic and membrane binding study. Brain Res Bull 1984; 13:657–66
Geiger JD, Nagy JI. Localization of [3H]nitrobenzylthioinosine binding sites in rat spinal cord and primary afferent neurones. Brain Res 1985; 347: 321–7
Sawynok J, Sweeney MI. The role of purines in nociception. Neuroscience 1989; 32: 557–69
Ahlijanian MK, Takemori AE. Effects of (−)-N6-(R-phenylisopropyl) -adenosine (PIA) and caffeine on nociception and morphine-induced analgesia, tolerance and dependence in mice. Eur J Pharmacol 1985; 112: 171–9
Holmgren M, Hedner J, Nordberg G, et al. Antinociceptive effects in the rat of an adenosine analogue, N6-phenylisopro-pyladenosine. J Pharm Pharmacol 1983; 35: 679–80
Holmgren M, Hedner J, Mellstrand T, et al. Characterization of the antinociceptive effects of some adenosine analogues in the rat. Naunyn Schmiedebergs Arch Pharmakol 1986; 334: 290–3
Vapaatalo H, Onken D, Neuvonen PJ, et al. Stereospecificity in some central and circulatory effects of phenylisopropyl-adenosine (PIA). Arzneimittl-Forsch 1975; 25: 407–10
Herrick-Davis K, Chippari S, Luttinger D, et al. Evaluation of adenosine agonists as potential analgesics. Eur J Pharmacol 1989; 162: 365–9
DeLander GE, Hopkins CJ. Spinal adenosine modulates descending antinociceptive pathways stimulated by morphine. J Pharm Exp Ther 1986; 239: 88–93
Doi T, Kuzuna S, Maki Y. Spinal antinociceptive effects of adenosine compounds in mice. Eur J Pharmacol 1987; 137: 227–31
Karlsten R, Gordh Jr T, Hartvig P, et al. Effects of intrathecal injection of the adenosine receptor agonists R-phenylisopropyl-adenosine and N-ethylcarboxamide-adenosine on nociception and motor function in the rat. Anesth Analg 1990; 71: 60–4
Malmberg AB, Yaksh TL. Pharmacology of the spinal action of ketorolac, morphine, ST-91, U50488H, and L-PIA on the formalin test and isobolographic analysis of the NSAID interaction. Anesthesiology 1993; 79: 270–81
Post C. Antinociceptive effects in mice after intrathecal injection of 5′-N-Ethylcarboxamide adenosine. Neurosci Lett 1984; 51: 325–30
Sawynok J, Sweeney MI, White TD. Classification of adenosine receptors mediating antinociception in the rat spinal cord. Br J Pharmacol 1986; 88: 923–30
Sosnowski M, Yaksh TL. Role of spinal adenosine receptors in modulating the hyperesthesia produced by spinal glycine receptor antagonism. Anesth Analg 1989; 69: 587–92
Yarbrough GY, McGuffm-Clineschmidt JC. In vivo behavioral assessment of central nervous system purinergic receptors. Eur J Pharmacol 1981; 76: 137–44
Reeve AJ, Dickenson AH. Electrophysiological study on spinal antinociceptive interactions between adenosine and morphine in the dorsal horn of the rat. Neurosci Lett 1995; 194: 81–4
Reeve AJ, Dickenson AH. The roles of spinal adenosine receptors in the control of acute and more persistent nociceptive responses of dorsal horn neurones in the anaesthetized rat. BrJ Pharmacol 1995; 116: 2221–8
Reeve AJ, Dickenson AH, Kerr NC. Spinal effects of bicuculline: modulation of an allodynia-like state by an A1-receptor agonist, morphine, and an NMDA-receptor antagonist, J Neurophys 1998; 79: 1494–507
Mendell LM. Physiological properties of unmyelinated fiber projection to the spinal cord. Exp Neurol 1966; 16: 316–32
Davies SN, Lodge D. Evidence for involvement of N-methyl-D-aspartate receptors in wind-up of class 2 neurones in the dorsal horn of the rat. Brain Res 1987; 424: 402–6
Dickenson AH, Sullivan AF. Evidence for a role of the NMDA receptor in the frequency dependent potentiation of deep rat dorsal horn nociceptive neurones following C-fibre stimulation. Neuropharmacol 1987; 26: 1235–8
White TD. Potentiation of excitatory amino-acid-evoked adenosine release from rat cortex by inhibitors of adenosine kinase and adenosine deaminase and by acadesine. Eur J Pharmacol 1996; 303: 27–38
Dora CD, Koch S, Sanchez A, et al. Intraspinal injection of adenosine agonists protect against L-NAME induced neuronal loss in the rat. J Neurotrauma 1998; 15: 473–83
Keil GJII, DeLander GE. Altered sensory behaviors in mice following manipulation of endogenous spinal adenosine neurotransmission. Eur J Pharmacol 1996; 312: 7–14
Backonja M, Arndt G, Gombar KA, et al. Response of chronic neuropathic pain syndromes to ketamine: apreliminary study. Pain 1994; 56: 51–7
Chapman V, Dickenson AH. Enhanced responses of rat dorsal horn neurones after UV irradiation of the hindpaw; roles of the NMDA receptor. Neurosci Lett 1994; 176: 41–4
Dickenson AH. NMDA receptor antagonists as analgesics. In: Fields HL, Liebeskind JC, editors. Pharmacological approaches to the treatment of chronic pain: new concepts and critical issues. Progress in pain research and management. Vol. 1. Seattle (WA): IASP Press, 1994: 173–87
Eide PK, Jorum E, Stubhaug A, et al. Relief of post-herpetic neuralgia with the N-methyl-D-aspartic acid receptor antagonist ketamine: a double-blind, cross-over comparison with morphine and placebo. Pain 1994; 58: 347–54
Ma Q-P, Woolf CJ. Noxious stimuli induce an N-methyl-D-aspartate receptor-dependent hypersensitivity of the flexion withdrawal reflex to touch: implications for the treatment of mechanical allodynia. Pain 1995; 61: 383–90
Persson J, Axelsson G, Hallin RG, et al. Beneficial effects of ketamine in a chronic pain state with allodynia, possibly due to central sensitization. Pain 1995; 60: 217–22
Sherman SE, Loomis CW. Morphine insensitive allodynia is produced by intrathecal strychnine in the lightly anesthetized rat. Pain 1994; 56: 17–29
Yaksh TL. Behavioral and autonomic correlates of the tactile evoked allodynia produced by spinal glycine inhibition: effects of modulatory receptor systems and excitatory amino acid antagonists. Pain 1989; 37: 111–23
Desmeules JA, Kayser V, Guilbaud G. Selective opioid receptor agonists modulate mechanical allodynia in an animal model of neuropathic pain. Pain 1993; 53: 277–85
Jadad AR, Carroll D, Glynn CJ, et al. Morphine responsiveness of chronic pain: double-blind randomized crossover study with patient-controlled analgesia. Lancet 1992; 339: 1367–71
Sollevi A. Adenosine for pain control. Acta Anaesthesiol Scand 1997;Suppl. 110: 135–6
Segerdahl M, Ekblom A, Sollevi A. The influence of adenosine, ketamine, and morphine on experimentally induced ischemic pain in healthy volunteers. Anesth Analg 1994; 79: 787–91
Segerdahl M, Ekblom A, Sjolund K-F, et al. Systemic adenosine attenuates touch evoked allodynia induced by mustard oil in humans. Neuroreport 1995; 6: 753–6
Ekblom A, Segerdahl M, Sollevi A. Adenosine increases the cutaneous heat pain threshold in healthy volunteers. Acta Anaesthesiol Scand 1995; 39: 717–22
Rane K, Segerdahl M, Goiny M, et al. Intrathecal adenosine administration: a phase 1 clinical safety study in healthy volunteers, with additional evaluation of its influence on sensory thresholds and experimental pain. Anesthesiology 1998; 89: 1108–15
Kingery WS. A critical review of controlled clinical trials for peripheral neuropathic pain and complex regional syndrome. Pain 1997; 73: 123–39
Guieu R, Peragut JC, Roussel P, et al. Adenosine and neuropathic pain. Pain 1996; 68: 271–4
Sugimoto T, Bennett GJ, Kajander KC. Transsynaptic degeneration in the superficial dorsal horn after sciatic nerve injury: effects of chronic constriction injury, transection, and strychnine. Pain 1990; 42: 205–13
Sjolund KF, von Heijne M, Hao JX, et al. Intrathecal administration of the adenosine A1 receptor agonist R-phenylisopropyl adenosine reduces presumed pain behaviour in a rat model of central pain. Neurosci Lett 1998; 243: 89–92
Cui J-G, Sollevi A, Linderoth B, et al. Adenosine receptor activation suppresses tactile hypersensitivity and potentiates spinal cord stimulation in mononeuropathic rats. Neurosci Lett 1997; 223: 173–6
Lee Y-W, Yaksh TL. Pharmacology of the spinal adenosine receptor which mediates the antiallodynic action of intrathecal adenosine agonists. J Pharmacol Exp Ther 1996; 277: 1642–8
Sjolund KF, Sollevi A, Segerdahl M, et al. Intrathecal and systemic R-phenylisopropyl-adenosine reduces scratching behaviour in a rat mononeuropathy model. Neuroreport 1996; 7: 1856–60
Kim SH, Chung JM. An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 1992; 50: 355–63
Lavand’homme PM, Eisenach JC. Exogenous and endogenous adenosine enhance the spinal antiallodynic effects of morphine in a rat model of neuropathic pain. Pain 1999; 80: 31–6
Beifrage M, Sollevi A, Segerdahl M, et al. Systemic adenosine infusion alleviates spontaneous and stimulus evoked pain in patients with peripheral neuropathic pain. Anesth Analg 1995; 81: 713–7
Sollevi A, Belfrage, Lundeberg T, et al. Systemic adenosine infusion: a new treatment modality to alleviate neuropathic pain. Pain 1995; 61: 155–8
Karlsten R, T Jr Gordh. An A1-selective adenosine agonist abolishes allodynia elicited by vibration and touch after intrathecal injection. Anesth Analg 1995; 80: 844–7
Sollevi A, Rane K, Segerdahl M, et al. Intrathecal adenosine injection elevates cutaneous pain thresholds in man [abstract]. In: 8th World Congress on Pain: 1996 Aug 18–22: Vancouver. Seattle (WA): IASP Press, 1996: 356
Dolphin AC, Prestwich SA. Pertussis toxin reverses adenosine inhibition of neuronal glutamate release. Nature 1985; 316: 148–50
Craig CG, White TD. Low-level N-methyl-D-aspartate receptor activation provides a purinergic inhibitory threshold against further N-methyl-D-aspartate-mediated neurotransmission in the cortex. J Pharm Exp Ther 1992; 260: 1278–84
Craig CG, White TD. N-methyl-D-aspartate and non-N-methyl D-aspartate-evoked adenosine release from rat cortical slices: distinct purinergic sources and mechanisms of release. J Neurochem 1993; 60: 1073–80
Conway CM, Marsala M, Somogyi GT, et al. Intrathecal NMDA-induced release of spinal adenosine and amino acids. Soc Neurosci Abstr 1997; 23: 1013
Mao J, Price DD, Mayer DJ, et al. Intrathecal MK-801 and local nerve anesthesia synergistically reduce nociceptive behaviors in rats with experimental peripheral mononeuropathy. Brain Res 1992; 576: 254–62
Qian J, Brown SD, Carlton SM. Systemic ketamine attenuates nociceptive behaviors in a rat model of peripheral neuropathy. Brain Res 1996; 715: 51–62
Sumida T, Smith MA, Maehara Y, et al. Spinal R-phenyl-isopropyl adenosine inhibits spinal dorsal horn neurons responding to noxious heat stimulation in the absence and presence of sensitization. Pain 1998; 74: 307–13
Sawynok J. Purines in pain management. Curr Opin CPNS Invest Drugs 1999; 1: 27–38
Dowd E, McQueen DS, Chessell IP, et al. Adenosine A1 receptor-mediatedexcitation of nociceptive afferents innervatingthe normal and arthritic knee joint. Br J Pharm 1998; 125:1267–71
Sawynok J, Zarrindast MR, Reid AR, et al. Adenosine A3 receptor activation produces nociceptive behaviour and edema by release of histamine and 5-hydroxytryptamine. Eur J Pharmacol 1997; 333: 1–7
Sawynok J. Adenosine receptor activation and nociception. Eur J Pharmacol 1998; 347: 1–11
Abbott FV, Melzack R, Samuel C. Morphine analgesia in the tail-flick and formalin pain tests is mediated by different neural systems. Exp Neurol 1982; 75: 644–51
Dubuisson D, Dennis SG. The formalin test: a quantitative study of the analgesic effects of morphine, meperidine, and brain stem stimulation in rats and cats. Pain 1977; 4: 161–74
Hargreaves K, Dubner R, Brown F, et al. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 1988; 32: 77–88
Tjolsen A, Berge O-G, Hunskaar S, et al. The formalin test: an evaluation of the method. Pain 1992; 51: 5–17
Wheeler-Aceto H, Cowan A. Standardization of the rat paw formalin test for the evaluation of analgesics. Psychopharmacol 1991; 104: 35–44
Doak GJ, Sawynok J. Complex role of peripheral adenosine in the genesis of the response to subcutaneous formalin in the rat. Eur J Pharmacol 1995; 281: 311–8
Karlsten R, Gordh T, Post C. Local antinociceptive and hyperalgesic effects in the formalin test after peripheral administration of adenosine analogues in mice. Pharmacol Toxicol 1992; 70: 434–8
Holton FA, Holton P. The capillary dilator substances in dry powders of spinal roots: a possible role of adenosine triphosphate in chemical transmission from nerve endings. J Physiol 1954; 126: 124–40
Holton P. The liberation of adenosine triphosphate on antidromic stimulation of sensory nerves. J Physiol 1959; 145:494–504
Rae CP, Mansfield MD, Dryden C, et al. Analgesic effect of adenosine on ischaemic pain in human volunteers. Br J Anaesth 1999; 82: 427–8
Sjolund KF, Segerdahl M, Sollevi A. Adenosine reduces secondary hyperalgesia in two human models of cutaneous inflammatory pain. Anesth Analg 1999; 88: 605–10
Bleehen T, Keele CA. Observations on the allogenic actions of adenosine compounds on the human blister base preparation. Pain 1977; 3: 367–77
Poon A, Sawynok J. Antinociception by adenosine analogs and an adenosine kinase inhibitor: dependence on formalin concentration. Eur J Pharmacol 1995; 286: 177–84
Sawynok J, Reid A, Poon A. Peripheral antinociceptive effect of an adenosine kinase inhibitor, with augmentation by an adenosine deaminase inhibitor, in the rat formalin test. Pain 1998; 74: 75–81
Keil 2nd GJ, DeLander GE. Spinally-mediated antinociception is induced in mice by an adenosine kinase-,but not by an adenosine deaminase-,inhibitor. Life Sci 1992; 51: 171–6
Keil 2nd GJ, DeLander GE. Adenosine kinase and adenosine deaminase inhibition modulate spinal adenosine- and opioid agonist-induced antinociception in mice. Eur J Pharmacol 1994; 271: 37–46
Poon A, Sawynok J. Antinociception by adenosine analogues and inhibition of adenosine metabolism in an inflammatory thermal hyperalgesia model in the rat. Pain 1998; 74: 235–45
Meghji P. Adenosine production and metabolism. In: Stone TW, editor. Adenosine in the nervous system. London: Academic Press, 1991: 25–42
Dickenson AH, Sullivan AF. Combined therapy in analgesia; seeking synergy. Curr Opin Anaesthesiol 1993; 6: 861–5
DeLander GE, Hopkins CJ. Involvement of A2 adenosine receptors in spinal mechanisms of antinociception. Eur J Pharm 1987; 139: 215–23
Segerdahl M, Forsberg C, Sollevi A. Ketamine and adenosine infusion prevent thermal changes in the secondary hyperalgesic area after mustard oil skin application [abstract]. In: 8th World Congress on Pain: 1996 Aug 18–22: Vancouver. Seattle (WA): IASP Press, 1996: 35
Sollevi A. Adenosine infusion during isoflurane-nitrous oxide anaesthesia: indications of perioperative analgesic effect. Acta Anaesthesiol Scand 1992; 36: 595–9
Sylven C, Eriksson B, Jensen J, et al. Analgesic effects of adenosine during exercise-provoked myocardial ischaemia. Neuroreport 1996; 7: 1521–5
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dickenson, A.H., Suzuki, R. & Reeve, A.J. Adenosine as a Potential Analgesic Target in Inflammatory and Neuropathic Pains. Mol Diag Ther 13, 77–85 (2000). https://doi.org/10.2165/00023210-200013020-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00023210-200013020-00001