Abstract
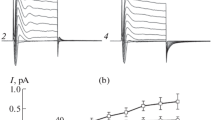
The possible role of Cl− currents in regulatory volume decrease processes has been explored in HeLa cells using the whole-cell recording mode of the patch-clamp technique. Cells showed very small currents in voltage-clamp experiments performed with Cl−-rich, permeant-cation-free (N-methyl-d-glucamine replacement) intracellular and bathing solutions. Exposure of the cells to hypotonic solutions visibly swelled the cells and activated, reversibly, an outward rectifying Cl− current, which decayed at the most depolarised voltages used. Replacement of extracellular Cl− by a series of halide anions, SCN− and gluconate was consistent with an anion selectivity sequence: SCN−>I−>Br−>Cl− >F−>gluconate. The volume-regulated Cl− current was effectively inhibited by 100 μM 5-nitro-2-(3-phenylpropylamino)-benzoic acid and by 100 μM 4,4′-diisothiocyanotostilbene-2,2-disulphonic acid, substances known to block Cl− channels in a variety of cells. Chloride current activation by hypotonicity was dependent on the presence of ATP in the intracellular solution and this requirement could be replaced by the non-hydrolysable analogue ATP[γS] and Mg2+-free ATP. The data suggest that the channels responsible for the current described are involved in the regulatory volume decrease in HeLa cells. The characteristics of this Cl− current are similar to those of the current associated with expression of multidrug resistance P-glycoprotein. Furthermore, the currents in HeLa cells were inhibited rapidly and reversibly by verapamil and 1,9-dideoxyforskolin, which are known to inhibit P-glycoprotein function.
Similar content being viewed by others
References
Bridges RJ, Worrell RT, Frizzell RA, Benos DJ (1989) Stilbene disulfonate blockade of colonic secretory Cl− channels in planar lipid bilayers. Am J Physiol 256:C902-C912
Champigny G, Verrier B, Lazdunski M (1990) Ca2+ channel blockers inhibit secretory Cl− channels in intestinal epithelial cells. Biochem Biophys Res Commun 171:1022–1028
Christenson O (1987) Mediation of cell volume regulation by Ca2+ influx through stretch-activated channels. Nature 330: 66–68
Frizzell RA, Halm D (1990) Chloride channels in epithelial cells. In: Helman SI, Van Driessche W (eds) Current topics in membrane and transport. Academic Press, New York, pp 247–282
Giraldez F, Valverde MA, Sepúlveda FV (1988) Hypotonicity increases apical membrane Cl− conductance in Necturus enterocytes. Biochim Biophys Acta 942:353–356
Giraldez F, Murray KJ, Sepúlveda FV, Sheppard DN (1989) Characterization of a phosphorylation-activated Cl-selective channel in isolated Necturus enterocytes. J Physiol (Lond) 416:517–537
Hahn DR, Rechkemmer GR, Schoumacher RA, Frizzell RA (1988) Apical membrane chloride channels in a chloride cell line activated by secretory agonists. Am J Physiol 254:C505-C511
Hazama A, Okada Y (1988) Ca2+ sensitivity of volume-regulatory K+ and Cl− channels in cultured human epithelial cells. J Physiol (Lond) 402:687–702
Hazama A, Okada Y (1990) Biphasic rises in cytosolic free Ca2+ in association with activation of K+ andCl− conductance during the regulatory volume decrease in cultured human epithelial cells. Pflügers Arch 416:710–714
Hoffmann EK, Simonsen LO (1989) Membrane mechanisms in volume and pH regulation in vertebrate cells. Physiol Rev 69:315–373
Okada Y, Hazama A (1989) Volume-regulatory ion channels in epithelial cells. News Physiol Sci 4:238–242
Sachs F, Sokabe M (1990) The structure and dynamics of patch-clamped membranes: a study using differential interference contrast light microscopy. J Cell Biol 111:599–606
Sauve R, Simoneau C, Monette R, Roy G (1986) Single-channel analysis of the potassium permeability in HeLa cancer cells: evidence for a calcium-activated potassium channel of small unitary conductance. J Membr Biol 92:269–282
Sheppard DN, Valverde MA, Giraldez F, Sepúlveda FV (1991) Potassium currents of isolated Necturus enterocytes: a whole-cell patch-clamp study. J Physiol (Lond) 433:663–676
Solc CK, Wine JJ (1991) Swelling-induced and depolarization-induced Cl− channels in normal and cystic fibrosis epithelial cells. Am J Physiol 261:C658-C674
Suzuki S, Tachibana M, Kaneko A (1990) Effects of glycine and GABA on isolated bipolar cells of the mouse retina. J Physiol (Lond) 421:645–662
Tabcharani JA, Low W, Elie D, Hanrahan JW (1990) Low-conductance chloride channel activated by cAMP in the epithelial cell line T84. FEBS Lett 270:157–164
Tilmann M, Kunzelmann K, Frobe U, Cabantchik I, Lang HJ, Englert HC, Greger R (1991) Different types of blockers of the intermediate-conductance outwardly rectifying chloride channel in epithelia. Pflügers Arch 418:556–563
Tivey DR, Simmons NL, Aiton JF (1985) Role of passive potassium fluxes in cell volume regulation in cultured HeLa cells. J Membr Biol 87:93–105
Ubl J, Murer H, Kolb H-A (1988) Hypotonic shock evokes opening of Ca2+-activated K channels in opossum kidney cells. Pflügers Arch 412:551–553
Valverde MA, Díaz M, Sepúlveda FV, Gill DH, Hyde SC, Higgins CF (1992) Volume-regulated chloride channels associated with the multidrug resistance P-glycoprotein. Nature 355:830–833
Wagner JA, Cozens AL, Schulman H, Gruenert DC, Stryer L, Gardner P (1991) Activation of chloride channels in normal and cystic fibrosis airway epithelial cells by multifunctional calcium/calmodulin-dependent protein kinase. Nature 349:793–796
Worrell RT, Butt AG, Cliff WH, Frizzell RA (1989) A volume-sensitive chloride conductance in human colonic cell line T84. Am J Physiol 256:C1111-C1119
Yang X-C, Sachs F (1990) Characterization of stretch-activated ion channels in Xenopus oocytes. J Physiol (Lond) 431:103–122
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Díaz, M., Valverde, M.A., Higgins, C.F. et al. Volume-activated chloride channels in HeLa cells are blocked by verapamil and dideoxyforskolin. Pflügers Arch. 422, 347–353 (1993). https://doi.org/10.1007/BF00374290
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00374290