Summary
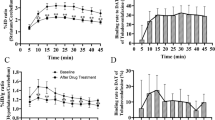
In NG 108-15 cells expressing the recombinant human D3 receptor, dopamine agonists enhance [3H]thymidine incorporation and decrease cAMP accumulation. In these cells, but not in wild type cells, haloperidol, fluphenazine, and various other antipsychotics inhibited basal [3H]thymidine incorporation in a concentration-dependent manner. In contrast, other dopamine antagonists such as nafadotride or (+)AJ 76, two D3-preferring antagonists, were without effect. The concentration-response curve of haloperidol was shifted to the right in presence of nafadotride, with a potency compatible with its nanomolar apparent affinity as neutral antagonist. Pertussis toxin treatment abolished or markedly reduced the responses to haloperidol or fluphenazine. In contrast, no significant enhancement of cAMP accumulation could be observed, under the influence of haloperidol or eticlopride. These data indicate that some dopamine antagonists behave as inverse agonists, and thus appear to inhibit an agonist-independent activity of the D3 receptor on [3H]thymidine incorporation pathway, but not on the cAMP pathway.
Similar content being viewed by others
References
Adie EJ, Milligan G (1994) Regulation of basal adenylate cyclase activity in neuroblastoma X glioma hybrid, NG 108-15, cells transfected to express the human α2 adrenoceptor: evidence for empty receptor stimulation of the adenylate cyclase cascade. Biochem J 303: 803–808
Barker EL, Westphal RS, Schmidt D, Sanders-Bush E (1994) Constitutively active 5-hydroxytryptamine 2c receptors reveal novel inverse agonist activity of receptors ligands. J Biol Chem 269: 11687–11690
Bond AR, P. Leff TD, Johnson CA, Milano HA, Rockman TR, McMinn S, Apparsundaram MF, Hyek TP, Kenakin LF, Allen LF, Lefkowitz RJ (1995) Physiological effects of inverse agonists in transgenic mice with myocardial overexpression of the α2-adrenoreceptor. Nature 374: 272–276
Boone C, Davis NG, Sprague GF (1993) Mutations that alter the third cytoplasmic loop of the a-factor receptor lead to a constitutive and hypersensitive phenotype. Proc Natl Acad Sci USA 90: 9921–9925
Chidiac P, Hebert TE, Valiquette M, Dennis M, Bouvier M (1994) Inverse agonist activity of β-adrenergic antagonists. Mol Pharmacol 45: 490–499
Chio CL, Lajiness ME, Huff RM (1994) Activation of heterologously expressed D3 dopamine receptors: comparison with D2 dopamine receptors. Mol Pharmacol 45: 51–60
Costa TH, Herz A (1989) Antagonists with negative intrinsic activity at opioid receptors coupled to GTP-binding proteins. Proc Natl Acad Sci USA 86: 7321–7325
Costa T, Ogino Y, Munson PJ, Oranan HO, Rodbard D (1992) Drug efficacy at guanine nucleotide-binding regulatory protein linked receptors: thermodynamic interpretation of negative antagonism and of receptor activity in the absence of ligand. Mol Pharmacol 41: 549–560
Cotecchia S, Exum S, Caron MG, Lefkowitz RJ (1990) Regions of the α1-adrenergic receptor involved in coupling to phosphatidylinositol hydrolysis and enhanced sensitivity of biological function. Proc Natl Acad Sci USA 87: 2896–2900
DeLean A, Stadel JM, Lefkowitz RJ (1980) A ternary complex model explains the agonist-specific binding properties of the adenylate cyclase-coupled β-adrenergic receptor. J Biol Chem 255: 7108–7117
Diaz J, Lévesque D, Lammers CH, Griffon N, Martres M-P, Schwartz J-C, Sokoloff P (1995) Phenotypical characterization of neurons expressing the dopamine D3 receptor. Neuroscience 65: 731–745
Duprez L, Parma J, Van Sande J, Allgeier A, Leclère J, Schvartz C, Deliste MJ, Decoulx M, Orgiazzi J, Dunont J, Vassart G (1994) Germline mutations in the tyrotropin receptor gene cause non-autoimmune autosomal dominant hyperthyroidism. Nature Genet 7: 396–401
Freedman SB, Patel S, Marwood R, Emms F, Seabrook GR, Knowles MR, McAllister G (1994) Expression and pharmacological characterization of the human D3 dopamine receptor. J Pharmacol Exp Ther 268: 417–426
Griffon N, Pilon C, Schwartz J-C, Sokoloff P (1995) The preferential dopamine D3 receptor ligand (+)UH 232 is a partial agonist. Eur J Pharmacol 282: R3-R4
Griffon N, Sautel F, Pilon C, Lévesque D, Sokoloff P, Schwartz J-C, Diaz J, Simon P, Costentin J, Mann A, Wermuth CG (1996) Functional models for the dopamine D3 receptor. Biochem Soc Trans 24: 193–198
Gurevich V, Kung MP, Bordelon Y, Joyce JN (1994) Dopamine D3 receptors are elevated in mesolimbic dopamine system of schizophrenics: receptor autoradiographic studies. Neuropsychopharmacol 10: 236S
Kjelsberg MA, Cotecchia S, Ostrowski J, Caron MG, Lefkowitz R (1992) Constitutive activation of the α1B-adrenergic receptor by all amino acid substitutions at a single site. Evidence for a region which constrains receptor activation. J Biol Chem 267: 1430–1433
Leff P (1995) The two-state model of receptor activation. Trends Pharmacol Sci 16: 89–97
MacKenzie RG, Van Leeuwen D, Pugsley TA, Shih YH, Demattos S, Tang L, Todd RD, O'Malley K (1994) Characterization of the human dopamine D3 receptor expressed in transfected cell lines. Eur J Pharmacol Mol Pharmacol Sect 266: 79–85
Milligan G, Bond RA, Lee M (1995) Inverse agonism: pharmacological curiosity or potential therapeutic strategy? Trends Pharmacol Sci 16: 10–13
Parma J, Duprez L, Van Sande J, Cochaux P, Gervy C, Mockel J, Dunont J, Vassart G (1993) Somatic mutations in the thyrotropin receptor gene cause hyperfunctioning thyroid adenomas. Nature 365: 649–651
Pilon C, Lévesque D, Dimitriadou V, Griffon N, Martres MP, Schwartz J-C, Sokoloff P (1994) Functional coupling of the human dopamine D3 receptor in a transfected NG 108-15 neuroblastoma-glioma hybrid cell line. Eur J Pharmacol [Mol Pharmacol Sect] 268: 129–139
Rao RV, Cohen GB, Oprian DD (1994) Rhodopsin mutation G90D and a molecular mechanism for congenital right blindness. Nature 367: 639–642
Ren Q, Kurose H, Lefkowitz RJ, Cotecchia S (1993) Constitutively active mutants of the alpha-2-adrenergic receptor. J Biol Chem 268: 16483–16487
Robbins LS, Nadeau JH, Johnson KR, Kelly MA, Roselli-Rehfuss L, Baack E, Mountjoy KG, Cone RD (1993) Pigmentation phenotypes of variant extension locus alleles result from point mutations that alter MSH receptor function. Cell 72: 827–834
Robinson PR, Cohen GB, Zhukovsky EA, Oprian DD (1992) Constitutively active mutants of rhodopsin. Neuron 9: 719–725
Samama P, Cotecchia S, Costa T, Lefkowitz RJ (1993) A mutation-induced activated state of the β2-adrenergic receptor. J Biol Chem 268: 4625–4636
Samama P, Pei G, Costa T, Cotecchia S, Lefkowitz RJ (1994) Negative antagonists promote an inactive conformation of the β2-adrenergic receptor. Mol Pharmacol 45: 390–394
Sautel F, Griffon N, Lévesque D, Pilon C, Schwartz JC, Sokoloff P (1995a) A functional test identifies dopamine agonists selective for D3 versus D2 receptors. NeuroReport 6: 329–332
Sautel F, Griffon N, Sokoloff P, Schwartz J-C, Launay C, Simon P, Costentin J, Schoenfelder A, Garrido F, Mann A, Wermuth CG (1995b) Nafadotride, a potent preferential dopamine D3 receptor antagonist, activates locomotion in rodents. J Pharmacol Exp Ther 275: 1239–1246
Seabrook GR, Patel S, Marwood R, Emms F, Knowles MR, Freedman SB, McAllister G (1992) Stable expression of human D3 dopamine receptor in GH4C1 pituitary cells. FEBS Lett 312: 123–126
Shenker A, Lane L, Kosugi S, Merendino JJ, Minegishi T, Cutler GB (1993) A constitutively activating mutation of the luteinizing hormone receptor in familial male precocious puberty. Nature 365: 652–654
Sokoloff P, Schwartz JC (1995) The novel dopamine receptors half a decade later. Trends Pharmacol Sci 16: 270–275
Sokoloff P, Giros B, Martres M-P, Bouthenet M-L, Schwartz J-C (1990) Molecular cloning and characterization of a novel dopamine receptor (D3) as a target for neuroleptics. Nature 347: 146–151
Sokoloff P, Andrieux M, Besançon R, Pilon C, Martres M-P, Giros B, Schwartz J-C (1992) Pharmacology of human D3 dopamine receptor expressed in a mammalian cell line: comparison with D2 receptor. Eur J Pharmacol Mol Pharmacol Sect 225: 331–337
Svensson K, Carlsson A, Huff RM, Kling-Petersen T, Waters N (1994) Behavioral and neurochemical data suggest functional differences between dopamine D2 and D3 receptors. Eur J Pharmacol 263: 235–243
Tang L, Todd RD, Heller A, O'Malley KL (1994) Pharmacological and functional characterization of D2, D3 and D4 dopamine receptors in fibroblasts and dopaminergic cell lines. J Pharmacol Exp Ther 268: 495–502
Tiberi M, Caron MG (1994) High agonist-independent activity is a distinguishing feature of the dopamine D1B receptor subtype. J Biol Chem 269: 27925–27931
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Griffon, N., Pilon, C., Sautel, F. et al. Antipsychotics with inverse agonist activity at the dopamine D3 receptor. J. Neural Transmission 103, 1163–1175 (1996). https://doi.org/10.1007/BF01271201
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01271201