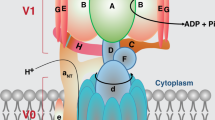
In this short review, we summarize our work on the role of members of the FXYD protein family as tissue-specific modulators of Na, K-ATPase. FXYD1 or phospholemman, mainly expressed in heart and skeletal muscle increases the apparent affinity for intracellular Na+ of Na, K-ATPase and may thus be important for appropriate muscle contractility. FXYD2 or γ subunit and FXYD4 or CHIF modulate the apparent affinity for Na+ of Na, K-ATPase in an opposite way, adapted to the physiological needs of Na+ reabsorption in different segments of the renal tubule. FXYD3 expressed in stomach, colon, and numerous tumors also modulates the transport properties of Na, K-ATPase but it has a lower specificity of association than other FXYD proteins and an unusual membrane topology. Finally, FXYD7 is exclusively expressed in the brain and decreases the apparent affinity for extracellular K+, which may be essential for proper neuronal excitability.
Similar content being viewed by others
References
Arystarkhova, E., Wetzel, R. K., Asinovski, N. K., and Sweadner, K. J. (1999). J. Biol. Chem. 274, 33183–33185.
Attali, B., Latter, H., Rachamim, N., and Garty, H. (1995). Proc. Natl. Acad. Sci. U.S.A. 92, 6092–6096.
Béguin, P., Crambert, G., Guennoun, S., Garty, H., Horisberger, J.-D., and Geering, K. (2001). EMBO J. 20, 3993–4002.
Béguin, P., Crambert, G., Monnet-Tschudi, F., Uldry, M., Horisberger, J.-D., Garty, H., and Geering, K. (2002). EMBO J. 21, 3264–3273.
Béguin, P., Wang, X. Y., Firsov, D., Puoti, A., Claeys, D., Horisberger, J. D., and Geering, K. (1997). EMBO J. 16, 4250–4260.
Chen, L.-S.K., Lo, C. F., Numann, R., and Cuddy, M. (1997). Genomics 41, 435–443.
Crambert, G., Fuzesi, M., Garty, H., Karlish, S., and Geering, K. (2002). Proc. Natl. Acad. Sci. U.S.A. 99, 11476–11481.
Crambert, G., and Geering, K. (2003). Sci. STKE 166, RE1.
Crambert, G., Hasler, U., Beggah, A. T., Yu, C., Modyanov, N. N., Horisberger, J. D., Lelievre, L., and Geering, K. (2000). J. Biol. Chem. 275, 1976–1986.
Crambert, G., Li, C., Claeys, D., and Geering, K. (2005). Mol. Biol. Cell 16, 2363–2371.
Crambert, G., Li, C., Swee, L. K., and Geering, K. (2004). J. Biol. Chem. 279, 30888–30895.
Feraille, E., and Doucet, A. (2001). Physiol. Rev. 81, 345–418.
Forbush, B. III, Kaplan, J. H., and Hoffman, J. F. (1978). Biochemistry 17, 3667–3676.
Fu, X., and Kamps, M. (1997). Mol. Cell. Biol. 17, 1503–1512.
Garty, H., Lindzen, M., Scanzano, R., Aizman, R., Fuzesi, M., Goldshleger, R., Farman, N., Blostein, R., and Karlish, S. J. D. (2002). Am. J. Physiol. 283, F607–F615.
Geering, K. (2001). J. Bioenerg. Biomembr. 33, 425–438.
Grzmil, M., Voigt, S., Thelen, P., Hemmerlein, B., Helmke, K., and Burfeind, P. (2004). Int. J. Oncol. 24, 97–105.
Hebert, H., Purhonen, P., Vorum, H., Thomsen, K., and Maunsbach, A. B. (2001). J. Mol. Biol. 314, 479–494.
Kowdley, G. C., Ackerman, S. J., Chen, Z. H., Szabo, G., Jones, L. R., and Moorman, J. R. (1997). Biophys. J. 72, 141–145.
Kuster, B., Shainskaya, A., Pu, H. X., Goldshleger, R., Blostein, R., Mann, M., and Karlish, S. J. (2000). J. Biol. Chem. 275, 18441–18446.
Li, C., Grosdidier, A., Crambert, G., Horisberger, J.-D., Michielin, O., and Geering, K. (2004). J. Biol. Chem. 279, 38895–38902.
Mahmmoud, Y. A., Cramb, G., Maunsbach, A. B., Cutler, C. P., Meischke, L., and Cornelius, F. (2003). J. Biol. Chem. 278, 37427–37438.
Maxwell, P. J., Longley, D. B., Latif, T., Boyer, J., Allen, W., Lynch, M., McDermott, U., Harkin, D. P., Allegra, C. J., and Johnston, P. G. (2003). Cancer Res. 63, 4602–4606.
Meij, I. C., Koenderink, J. B., van Bokhoven, H., Assink, K. F., Groenestege, W. T., de Pont, J. J., Bindels, R. J., Monnens, L. A., van den Heuvel, L. P., and Knoers, N. V. (2000). Nat. Genet. 26, 265–266.
Mercer, R. W., Biemesderfer, D., Bliss, D. P., Collins, J. H., and Forbush, B. (1993). J. Cell Biol. 121, 579–586.
Mirza, M. A., Zhang, X.-Q., Ahlers, B. A., Qureshi, A., Carl, L. L., Song, J., Tucker, A. L., Mounsey, J. P., Moorman, J. R., Rothblum, L. I., Zhang, T. S., and Cheung, J. Y. (2004). Am. J. Physiol. 286, H1322–H1330.
Moorman, J. R., Ackerman, S. J., Kowdley, G. C., Griffin, M. P., Mounsey, J. P., Chen, Z. H., Cala, S. E., Obrian, J. J., Szabo, G., and Jones, L. R. (1995). Nature 377, 737– 740.
Morrison, B. W., and Leder, P. (1994). Oncogene 9, 3417–3426.
Morrison, B. W., Moorman, J. R., Kowdley, G. C., Kobayashi, Y. M., Jones, L. R., and Leder, P. (1995). J. Biol. Chem. 270, 2176– 2182.
Palmer, C. J., Scott, B. T., and Jones, L. R. (1991). J. Biol. Chem. 266, 11126–11130.
Pu, H. X., Cluzeaud, F., Goldshlegger, R., Karlish, S. J. D., Farman, N., and Blostein, R. (2001). J. Biol. Chem. 276, 20370–20378.
Shi, H., Levy-Holzman, R., Cluzeaud, F., Farman, N., and Garty, H. (2001). Am. J. Physiol. 280, F505–F512.
Sweadner, K. J., and Rael, E. (2000). Genomics 68, 41–56.
Yamaguchi, F., Yamaguchi, K., Tai, Y., Sugimoto, K., and Tokuda, M. (2001). Brain Res. Mol. Brain Res. 86, 189–192.
Zhang, X.-Q., Qureshi, A., Song, J., Carl, L. L., Tian, Q., Stahl, R. C., Carey, D. J., Rothblum, L. I., and Cheung, J. Y. (2003). Am. J. Physiol. 284, H225–H233.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Geering, K. Function of FXYD Proteins, Regulators of Na, K-ATPase. J Bioenerg Biomembr 37, 387–392 (2005). https://doi.org/10.1007/s10863-005-9476-x
Issue Date:
DOI: https://doi.org/10.1007/s10863-005-9476-x