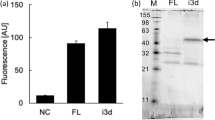
Abstract
We describe a procedure for isolating agonists for mammalian G protein–coupled receptors of unknown function. Human formyl peptide receptor like-1 (FPRL-1) receptor, originally identified as an orphan G protein–coupled receptor related to the formyl peptide receptor (FPR1), was expressed in Saccharomyces cells designed to couple receptor activation to histidine prototrophy. Selection for histidine prototrophs among transformants obtained with a plasmid-based library encoding random peptides identified six different agonists, each of whose production yielded autocrine stimulation of the receptor expressed in yeast. A synthetic version of each peptide promoted activation of FPRL-1 expressed in human embryonic kidney (HEK293) cells, and five of the peptides exhibited significant selectivity for activation of FPRL-1 relative to FPR1. One selective peptide was tested and found to mobilize calcium in isolated human neutrophils. This demonstrates that stimulation of FPRL-1 results in neutrophil activation and suggests that the receptor functions as a component of the inflammatory response. This autocrine selection protocol may be a generally applicable method for providing pharmacological tools to evaluate the physiological roles of the growing number of mammalian orphan G protein–coupled receptors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Devane, W.A., Hanuss, L., Breuer, A., Pertwee, R.G., Stevenson, L.A., Griffin, G. et al. 1992. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258: 1946–1949.
Meunier, J.-C., Mollereau C., Toll, L., Suaudeau, C., Moisand, C., Alvinerie, P. et al. 1995. Isolation and structure of the endogenous agonist of opioid receptor-like ORL-1 receptor. Nature 377: 532–535.
Murphy, P., Özçelik T., Kenney, R.T., Tiffany H.L., McDermott D., and Francke U. 1992. A structural homologue of the N-formyl peptide receptor. J. Biol. Chem. 267: 7637–7643.
Ye, R.D., Cavanagh, S.L., Quehenberger, O., Prossnitz, E.R., and Cochrane, C.G. 1992. Isolation of a cDNA that encodes a novel granulocyte N-formyl peptide receptor. Biochem. Biophys. Res. Commun. 184: 582– 589.
Durstin, M., Gao, J.-L., Tiffany, H.L., McDermott, D., and Murphy, P.M. 1994. Differential expression of members of the N-formylpeptide receptor gene cluster in human phagocytes. Biochem. Biophys. Res. Commun. 201: 174–179.
Bao, L., Gerard, N.P., Eddy R. Jr., Shows, T.B., and Gerard, C. 1992. Mapping of genes for the human C5a receptor (C5aR), human fMLP receptor (FPR), and two fMLP receptor homologue orphan receptors (FPRH1, FPRH2) to chromosome 19. Genomics 13: 437–440 .
Showell, H.J., Freer, R.J., Zigmond, S.H., Schiffman, E., Aswanikumar, S., Corcoran, B. et al. 1976. The structure-activity relations of synthetic peptides as chemotactic factors and inducers of lysosomal secretion for neutrophils. J. Exp. Med. 143: 1154–1169.
Fiore, S., Maddox, J.F., Perez, H.D., and Serhan, C.N. 1994. Identification of a human cDNA encoding a functional high affinity lipoxin A4 receptor. J. Exp. Med. 180: 253–260.
Manfredi, J.P., Klein, C., Herrero, J.J., Byrd, D.R., Trueheart, J., Wiesler, W.T. et al. 1996. Yeast a mating factor structure-activity relationship derived from genetically selected peptide agonists and antagonists of Ste2p. Mol. Cell. Biol. 16: 4700– 4709.
Stevenson, B.J., Rhodes, N., Errede, B., and Sprague, G.F. Jr. 1992. Constitutive mutants of the protein kinase STE11 activate the yeast pheromone response pathway in the absence of the G protein. Genes Dev. 6:1293–1304 .
Konteatis, Z.D., Siciliano, S.J., Van Riper, G., Molineaux, C.J., Pandya, S., Fischer, P. et al. 1994. Development of C5a receptor antagonists: differential loss of functional responses. J. Immunol. 153: 4200–4205.
Chen, J., Bernstein, H.S., Chen, M., Wang, L., Ishii, M., Turck, C.W. et al. 1995. Tethered ligand library for discovery of peptide agonists. J. Biol. Chem. 270: 23398–23401.
Romano, M., Maddox, J.F., and Serhan, C.N. 1996. Activation of human monocytes and the acute monocytic leukemia cell line (THP-1) by lipoxins involves unique signaling pathways for lipoxin A4 versus lipoxin B4. Evidence for differential Ca2+ mobilization. J. Immunol. 157: 2149–2154.
Libert, F., Parmentier, M., Lefort, A., Dinsart, C., Van Sande, J., Maenhaut, C. et al. 1989. Selective amplification and cloning of four new members of the G protein-coupled receptor family. Science 244: 569–572.
Feng, Y., Broder, C.C., Kennedy, P.E., and Berger, E.A. 1996. HIV-1 entry cofactor: functional cDNA cloning of a seven-transmembrane, G protein-coupled receptor. Science 272: 872–877.
Crass, T., Raffetseder, U., Martin, U., Grove, M., Klos, A., Köhl, J. et al. 1996. Expression cloning of the human C3a anaphylatoxin receptor (C3aR) from differentiated U-937 cells. Eur. J. Immunol. 26: 1944–1950.
Rose, A.B. and Broach, J.R. 1990. Propagation and expression of cloned genes in yeast: 2-micron circle-based vectors. Methods Enzymol. 185: 234–279.
Offermanns, S. and Simon, M.I. 1995 . G alpha 15 and G alpha 16 couple a wide variety of receptors to phospholipase C. J. Biol. Chem. 270: 15175 –15180.
Kaiser, C., Michaelis, S., and Mitchell, A. 1994. Methods in yeast genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York.
Kloptowski, T. and Wiater, A. 1965. Synergism of aminotriazole and phosphate on the inhibition of yeast imidazoleglycerolphosphate dehydratase. Arch. Biochem. Biophys. 112: 562–566.
Struhl, K. and Davis, R.W. 1977. Production of a functional eukaryotic enzyme in Escherichia coli: cloning and expression of the yeast structural gene for imidazoleglycerolphosphate dehydratase (his3). Proc. Natl. Acad. Sci. USA 74: 5255 –5259.
Acknowledgements
We thank Philip Murphy for providing plasmids encoding FPR and FPRL-1; William T. Wiesler for Cp1584 and β-galactosidase assays; Anna Farescal and Dwight Morrow for conducting peptide searches; Martin McKinney and Lorna Nuñez for technical assistance; and Nan-Xin Qian for providing the HEK293 cell line expressing Gα16. We also thank David R. Webb for a critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klein, C., Paul, J., Sauvé, K. et al. Identification of surrogate agonists for the human FPRL-1 receptor by autocrine selection in yeast. Nat Biotechnol 16, 1334–1337 (1998). https://doi.org/10.1038/4310
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/4310
This article is cited by
-
Formyl peptide receptor 2 as a potential therapeutic target for inflammatory bowel disease
Acta Pharmacologica Sinica (2023)
-
Functional expression of opioid receptors and other human GPCRs in yeast engineered to produce human sterols
Nature Communications (2022)
-
Designer cells programming quorum-sensing interference with microbes
Nature Communications (2018)
-
Effect of Annexin V on Development of Apoptosis in Placental Syncytiotrophoblast Nuclei during Exacerbation of Cytomegalovirus Infection in the Third Trimester
Bulletin of Experimental Biology and Medicine (2016)
-
High-throughput de novo screening of receptor agonists with an automated single-cell analysis and isolation system
Scientific Reports (2014)