Abstract
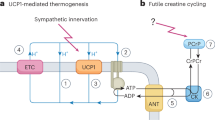
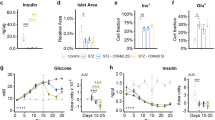
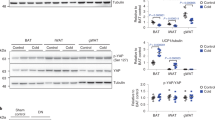
To determine whether uncoupling respiration from oxidative phosphorylation in skeletal muscle is a suitable treatment for obesity and type 2 diabetes, we generated transgenic mice expressing the mitochondrial uncoupling protein (Ucp) in skeletal muscle. Skeletal muscle oxygen consumption was 98% higher in Ucp-L mice (with low expression) and 246% higher in Ucp-H mice (with high expression) than in wild-type mice. Ucp mice fed a chow diet had the same food intake as wild-type mice, but weighed less and had lower levels of glucose and triglycerides and better glucose tolerance than did control mice. Ucp-L mice were resistant to obesity induced by two different high-fat diets. Ucp-L mice fed a high-fat diet had less adiposity, lower levels of glucose, insulin and cholesterol, and an increased metabolic rate at rest and with exercise. They were also more responsive to insulin, and had enhanced glucose transport in skeletal muscle in the setting of increased muscle triglyceride content. These data suggest that manipulating respiratory uncoupling in muscle is a viable treatment for obesity and its metabolic sequelae.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Saraste, M. Oxidative phosphorylation at the fin de siècleM. Science 283, 1488–1493 ( 1999).
Klingenberg, M. & Winkler, E. The reconstituted isolated uncoupling protein is a membrane potential-driven H+ translocator . EMBO J. 4, 3087–3092 (1985).
Enerback, S. et al. Mice lacking mitochondrial uncoupling protein are cold-sensitive but not obese. Nature 387, 90– 94 (1997).
Ravussin, E. et al. Reduced rate of energy expenditure as a risk factor for body-weight gain. N. Engl. J. Med. 318, 467– 472 (1988).
Holloszy, J.O. & Booth, F.W. Biochemical adaptations to endurance exercise in muscle. Ann. Rev. Physiol. 38, 273–291 (1976).
Boss, O., Hagen, T. & Lowell, B.B. Uncoupling proteins 2 and 3: Potential regulators of mitochondrial energy metabolism. Diabetes 49, 143–156 (2000).
Chung, W.K. et al. Genetic and physiologic analysis of the role of uncoupling protein 3 in human energy homeostasis. Diabetes 48, 1890–1895 (1999).
Argyropoulos, G. et al. Effects of mutations in the human uncoupling protein 3 gene on the respiratory quotient and fat oxidation in severe obesity and type 2 diabetes. J. Clin. Invest. 102, 1345– 1351 (1998).
Gong, D-W. et al. Lack of obesity and normal response to fasting and thyroid hormone in mice lacking uncoupling protein-3. J. Biol. Chem. 275, 16251–16257 (2000).
Vidal-Puig, A.J. et al. Energy metabolism in uncoupling protein 3 knockout mice. J. Biol. Chem. 275, 16258–16266 (2000).
Rial, E. et al. Retinoids activate proton transport by the uncoupling proteins UCP1 and UCP2. EMBO J. 18, 5827– 5833 (1999).
Cline, G.W. et al. Impaired glucose transport as a cause of decreased insulin-stimulated muscle glycogen synthesis in type 2 diabetes. N. Engl. J. Med. 341, 240–246 ( 1999).
Mueckler, M. Facilitative glucose transporters. Eur. J. Biochem. 219, 713–725 (1994).
Taylor, B.A. & Phillips, S.J. Detection of obesity QTLs on mouse chromosomes 1 and 7 by selective DNA pooling. Genomics 34, 389–398 ( 1996).
Tataranni, P.A. & Ravussin, E. Variability in metabolic rate: biological sites of regulation. Int. J. Obesity 19, S102–S106 ( 1995).
Rolfe, D.F., Newman, J.M., Buckingham, J.A., Clark, M.G. & Brand, M.D. Contribution of mitochondrial proton leak to respiration rate in working skeletal muscle and liver and to SMR. Am. J. Physiol. 276, C692– C699 (1999).
Ross, R. et al. Reduction in obesity and related comorbid conditions after diet-induced weight loss or exercise-induced weight loss in men. A randomized, controlled trial. Ann. Intern. Med. 133, 92– 103 (2000).
Portillo, M.P., Cantoral, R. & Macarulla, M.T. Effects of dietary fat content on adiposity during energy restriction in genetically obese rats. Reprod. Nutr. Dev. 39, 189–199 ( 1999).
Lowell, B.B. et al. Development of obesity in transgenic mice after genetic ablation of brown adipose tissue. Nature 366, 740 –742 (1993).
Klaus, S., Munzberg, H., Truloff, C. & Heldmaier, G. Physiology of transgenic mice with brown fat ablation: obesity is due to lowered body temperature. Am. J. Physiol. 274, R287 –R293 (1998).
Kopecky, J., Clark, G., Enerback, S., Spiegelman, B. & Kozak, L.P. Expression of the mitochondrial uncoupling protein gene from the aP2 gene promoter prevents genetic obesity. J. Clin. Invest. 96, 2914–2923 (1995).
Kopecky, J., Hodny, Z., Rossmeisl, M., Syrovy I. & Kozak, L.P. Reduction of dietary obesity in aP2-Ucp transgenic mice: physiology and adipose tissue distribution . Am. J. Physiol. E768–E775 (1996).
van den Ouweland, J.M.W. et al. Mutation in mitochondrial tRNA Leu(UUR) gene in a large pedigree with maternally transmitted type II diabetes mellitus and deafness. Nature Genetics 1, 368–371 (1992).
Gerbitz, K.-D., Gempel, K. & Brdiczka, D. Genetic, biochemical, and clinical implications of the cellular energy circuit. Diabetes 45, 113–126 (1996).
Poulton, J., Brown, M.S., Cooper, A., Marchington, D.R. & Phillips, D.I.W. A common mitochondrial DNA variant is associated with insulin resistance in adult life. Diabetologia 41, 54–58 ( 1998).
Huang, X. et al. Insulin-regulated mitochondrial gene expression is associated with glucose flux in human skeletal muscle. Diabetes 48, 1508–1524 (1999).
Holmes, B.F., Kurth-Kraczek, E.J. & Winder, W.W. Chronic activation of 5′-AMP-activated protein kinase increases GLUT-4, hexokinase, and glycogen in skeletal muscle. J. Appl. Physiol. 87, 1990–1995 (1999).
Hardie, D.G. Regulation of fatty acid and cholesterol metabolism by the AMP-activated protein kinase. Biochim. Biophys. Acta 1123, 231 –238 (1992).
Hansen, P.A. et al. A high fat diet impairs stimulation of glucose transport in muscle. J. Biol. Chem. 273, 26157– 26163 (1998).
Han, D.-H., Hansen, P.A., Host, H.H. & Holloszy, J.O. Insulin resistance of muscle glucose transport in rats fed a high-fat diet . Diabetes 46, 1761–1767 (1997).
Perseghin, G. et al. Intramyocellular triglyceride content is a determinant of in vivo insulin resistance in humans—A 1H-13C nuclear magnetic resonance spectroscopy assessment in offspring of type 2 diabetic parents. Diabetes 48, 1600–1606 (1999).
Hoppeler, H. & Weibel, E.R. Structural and functional limits for oxygen supply to muscle. Acta Physiol. Scand. 168, 445–56 (2000).
Seip, R.L. & Semenkovich, C.F. Skeletal muscle lipoprotein lipase: molecular regulation and physiological effects in relation to exercise. Exer. Sports Sci. 26, 191– 218 (1998).
Esposito, L.A., Melov, S., Panov, A., Cottrell, B.A. & Wallace, D.C. Mitochondrial disease in mouse results in increased oxidative stress. Proc. Natl. Acad. Sci. USA 96, 4820–4825 ( 1999).
Lee, C-K., Klopp, R.G., Weindruch, R. & Prolla, T.A. Gene expression profile of aging and its retardation by caloric restriction . Science 285, 1390–1393 (1999).
Beckman, K.B & Ames, B.N. Mitochondrial aging: open questions . Ann. NY Acad. Sci. 854, 118– 127 (1998).
Li, B., Holloszy, J.O. & Semenkovich, C.F. Respiratory uncoupling induces - aminolevulinate synthase expression through a nuclear respiratory factor-1-dependent mechanism in HeLa cells. J. Biol. Chem. 274, 17534– 17540 (1999).
Marshall, B.A. et al. Germline manipulation of glucose homeostasis via alteration of glucose transporter levels in skeletal muscle. J. Biol. Chem. 268, 18442–18445 ( 1993).
Scott, M.D., Baudendistel, L.J. & Dahms, T.E. Rapid separation of creatine, phosphocreatine and adenosine metabolites by ion-pair reversed-phase high performance liquid chromatography in plasma and cardiac tissue. J. Chromatography 576 , 149–154 (1992).
Uyeda, K. & Racker, E. Regulatory mechanisms in carbohydrate metabolism. VII. Hexokinase and phosphofructokinase. J. Biol. Chem. 240, 4682–4688 (1965).
Coleman, T., Seip, R.L., Gimble, J.M., Lee, D., Maeda, N. & Semenkovich, C.F. COOH-terminal disruption of lipoprotein lipase in mice is lethal in homozygotes, but heterozygotes have elevated triglycerides and impaired enzyme activity. J. Biol. Chem. 270, 12518–12525 (1995).
Marshall, B.A. et al. Relative hypoglycemia and hyperinsulinemia in mice with heterozygous lipoprotein lipase (LPL) deficiency. J. Biol. Chem. 274, 27426–27432 (1999).
Towler, D.A., Bidder, M., Latifi, T., Coleman, T. & Semenkovich, C.F. Diet-induced diabetes activates an osteogenic gene regulatory program in the aortas of low density lipoprotein receptor-deficient mice. J. Biol. Chem. 273, 30427– 30434 (1998).
Dobush, G.R., Ankey, C.D. & Kremantz, D.G. The effect of apparatus, extraction time, and solvent type on lipid extractions of snow geese. Can. J. Zool. 63, 1917–1920 (1985).
Acknowledgements
This work was supported by grants from the National Institutes of Health (HL58427, DK53198, AG00425 and AG14658), the Washington University Clinical Nutrition Research Unit (DK56341) and the Washington University Diabetes Research and Training Center (DK20579).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, B., Nolte, L., Ju, JS. et al. Skeletal muscle respiratory uncoupling prevents diet-induced obesity and insulin resistance in mice. Nat Med 6, 1115–1120 (2000). https://doi.org/10.1038/80450
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/80450
This article is cited by
-
Filbertone, (2E)-5-methyl-2-hepten-4-one, regulates thermogenesis and lipid metabolism in skeletal muscle of a high-fat diet fed mice
Applied Biological Chemistry (2023)
-
Loss of Lkb1 in CD11c+ myeloid cells protects mice from diet-induced obesity while enhancing glucose intolerance and IL-17/IFN-γ imbalance
Cellular and Molecular Life Sciences (2023)
-
Thermoreceptor TRPV1 regulates body weight and blood pressure in the absence of thermogenin
Hypertension Research (2022)
-
Non-alcoholic fatty liver disease and liver secretome
Archives of Pharmacal Research (2022)
-
Ccr7 null mice are protected against diet-induced obesity via Ucp1 upregulation and enhanced energy expenditure
Nutrition & Metabolism (2019)