Abstract
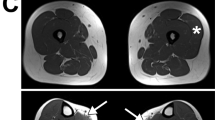
Limb-girdle muscular dystrophy (LGMD) is a clinically and genetically heterogeneous group of myopathies, including autosomal dominant and recessive forms1–3. To date, two autosomal dominant forms have been recognized2,3: LGMD1A, linked to chromosome 5q, and LGMD1B, associated with cardiac defects and linked to chromosome 1q11–21. Here we describe eight patients from two different families with a new form of autosomal dominant LGMD, which we propose to call LGMD1C, associated with a severe deficiency of caveolin-3 in muscle fibres. Caveolin-3 (or M-caveolin) is the muscle-specific form of the caveolin protein family, which also includes cave-olin-1 and -2 (refs 4–9). Caveolins are the principal protein components of caveolae (50–100 nm invaginations found in most cell types) which represent appendages or sub-compartments of plasma membranes10,11. We localized the human caveolin-3 gene (CAV3) to chromosome 3p25 and identified two mutations in the gene: a missense mutation in the membrane-spanning region and a micro-deletion in the scaffolding domain. These mutations may interfere with caveolin-3 oligomerization and disrupt caveolae formation at the muscle cell plasma membrane.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bushby, K. Towards the classification of the autosomal recessive limb-girdle muscular dystrophies. Neuromuscul. Disord. 6, 439–441 (1996).
Speer, M.C. et al. Confirmation of genetic heterogeneity in limb-girdle muscular dystrophy: linkage of an autosomal dominant form to chromosome 5q. Am. J. Hum. Genet. 50, 1211–1217 (1992).
Van der Kooi, A.J. et al. Genetic localization of a newly recognized autosomal dominant limb-girdle muscular dystrophy with cardiac involvement (LGMD 1B) to chromosome 1q11–21. Am. J. Hum. Genet. 60, 891–895 (1997).
Way, M. & Parton, R.G., caveolin, M.-, a muscle-specific caveolin-related protein. FEBS Lett. 376, 108–112 (1995).
Tang, Z. et al. Molecular cloning of caveolin-3, a novel member of the caveolin gene family expressed predominantly in muscle. J. Biol. Chem. 271, 2255–2261 (1996).
Parton, R.G. Caveolae and caveolins. Curr. Opin. Cell Biol. 8, 542–548 (1996).
Couet, J., Shengwen, L., Okamoto, T., Scherer, P.E. & Lisanti, M.P. Molecular and cellular biology of caveolae: Paradoxes and plasticities. Trends Cardiovasc. Med. 7, 103–110 (1997).
Scherer, P.E. et al. Identification, sequence, and expression of caveolin-2 defines a caveolin gene family. Proc. Natl. Acad. Sci. USA 93, 131–135 (1996).
Tang, Z. et al. Identification, sequence and expression of an invertebrate caveolin gene family from the Nematode Caenorhabditis elegans. J. Biol. Chem. 272, 2437–2445 (1997).
Yamada, E. The fine structure of the gall bladder epithelium of the mouse. J. Biophys. Biochem. Cytol. 1, 445–458 (1955).
Simionescu, N., Simionescu, M. & Palade, G.E. Permeability of muscle capillaries to small heme-peptides: evidence for the existence of patent transendothelial channels. J. Cell. Biol. 64, 586–607 (1975).
Couet, J., Li, S., Okamoto, T., Ikezu, T. & Lisanti, M.P. Identification of peptide and protein ligands for the caveolin-scaffolding domain: Implications for the interaction of caveolin with caveolae-associated proteins. J. Biol. Chem. 272, 6525–6533 (1997).
Okamoto, T., Schlegel, A., Scherer, P.E. & Lisanti, M.P., Caveolins, a family of scaffolding proteins for organizing ‘pre-assembled signaling complexes’ at the plasma membrane. J. Biol. Chem. in press.
Song, K.S. et al. Expression of caveolin-3 in skeletal, cardiac and smooth muscle cells: caveolin-3 is a component of the sarcolemma and co- fractionates with dystrophin and dystrophin-associated glycoproteins. J. Biol. Chem. 271, 15160–15165 (1996).
Hoffman, E.P., Brown, R.H. & Kunkel, L.M. Dystrophin: the protein product of the Duchenne muscular dystrophy. Cell 51, 919–928 (1987).
Ervasti, J.M. & Campbell, K.P. Membrane organization of the dystrophin-glycoprotein complex. Cell 66, 1121–1131 (1996).
Helbling-Leclerc, A. et al. Mutations in the laminin α2-chain (LAMA2) cause merosin-deficient congenital muscular dystrophy. Nature Genet. 11, 216–216 (1995).
Song, K.S., Tang, Z.L., Li, S. & Lisanti, M.P. Mutational analysis of the properties of caveolin-1: a novel role for the C-terminal domain in mediating homo-typing caveolin-caveolin interactions. J. Biol. Chem. 272, 398–403 (1997).
Li, S. et al. Evidence for a regulated interaction between heterotrimeric G proteins and caveolin. J. Biol. Chem. 270, 15693–15701 (1995).
Neer, E.J. Heterotrimeric G proteins: organizers of transmembrane signals. Cell 80, 249–257 (1995).
Song, K.S., Li, S., Okamoto, T., Quilliam, L., Sargiacomo, M. & Lisanti, M.P. Co-purification and direct interaction of Ras with caveolin, an integral membrane protein of caveolae microdomains: Detergent-free purification of caveolae membranes. J. Biol. Chem. 271, 9690–9697 (1996).
Li, S., Cuet, J. & Lisanti, M.P. Src tyrosin kinases, Gcc subunits and H-ras share a common membrane-anchored scaffolding protein, caveolin: caveolin binding negatively regulates the autoactivation of Src tyrosine kinases. J. Biol. Chem. 271, 29182–29190 (1996).
Couet, J., Sargiacomo, M. & Lisanti, M.P. Interaction of a receptor tyrosine kinase, EGF-R, with caveolins: Caveolin-binding negatively regulates tyrosine and serine/threonine kinase activities. J. Biol. Chem. 272, 30429–30438 (1997).
Oka, N. et al. Caveolin interaction with protein kinase C: Isoenzyme-dependent regulation of kinase activity by the caveolin-scaffolding domain peptide. J. Biol. Chem. 272, 33416–33421 (1997).
Feron, O., Belhassen, L., Kobzik, L., Smith, T.W., Kelly, R.A. & Michel, T. Endothelial nitric oxide synthase targeting to caveolae. J. Biol. Chem. 271, 22810–22814 (1996).
Garcia-Cardena, G. et al. Dissecting the interaction between nitric oxide synthase (NOS) and caveolin: Functional significance of the NOS caveolin binding domain in vivo. J. Biol. Chem. 272, 25437–25440 (1997).
Scherer, P.S. et al. Caveolin isoforms differ in their N-terminal protein sequence and subcellular distribution: Identification and epitope mapping of an isoform-specific monoclonal antibody probe. J. Biol. Chem. 270, 16395–16401 (1995).
Scherer, P.E. et al. Cell-type and tissue-specific expression of caveolin-2: Caveolins 1 and 2 co-localize and form a stable hetero-oligomeric complex in vivo. J. Biol. Chem. 272, 29337–29346 (1997).
Minetti, C. et al. Abnormalities in the expression of β-spectrin in Duchenne muscular dystrophy. Neurology 44, 1149–1153 (1994).
Scartezzini, P. et al. Cloning a new human gene from chromosome 21q22.3 encoding a glutamic acid-rich protein expressed in heart and skeletal muscle. Hum. Genet. 99, 387–392 (1997).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Minetti, C., Sotgia, F., Bruno, C. et al. Mutations in the caveolin-3 gene cause autosomal dominant limb-girdle muscular dystrophy. Nat Genet 18, 365–368 (1998). https://doi.org/10.1038/ng0498-365
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0498-365
This article is cited by
-
Loss of dysferlin or myoferlin results in differential defects in excitation–contraction coupling in mouse skeletal muscle
Scientific Reports (2021)
-
Caveolin 1 is required for axonal outgrowth of motor neurons and affects Xenopus neuromuscular development
Scientific Reports (2020)
-
Defective membrane repair machinery impairs survival of invasive cancer cells
Scientific Reports (2020)
-
Endothelial caveolin and its scaffolding domain in cancer
Cancer and Metastasis Reviews (2020)
-
Caveolin1 Tyrosine-14 Phosphorylation: Role in Cellular Responsiveness to Mechanical Cues
The Journal of Membrane Biology (2020)