Abstract
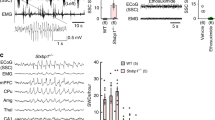
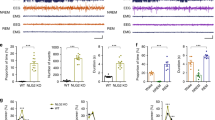
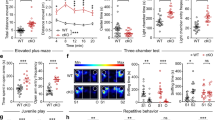
Absence epilepsy is a neurological disorder that causes a recurrent loss of consciousness and generalized spike-and-wave discharges on an electroencephalogram (EEG). The role of metabotropic glutamate receptors (mGluRs) and associated scaffolding proteins in absence epilepsy has been unclear to date. We investigated a possible role for these proteins in absence epilepsy, focusing on the mGluR7a receptor and its PDZ-interacting protein, protein interacting with C kinase 1 (PICK1), in rats and mice. Injection of a cell-permeant dominant-negative peptide or targeted mutation of the mGluR7a C terminus, both of which disrupt the interaction between the receptor and PDZ proteins, caused behavioral symptoms and EEG discharges that are characteristic of absence epilepsy. Inactivation of the Pick1 gene also facilitated pharmacological induction of the absence epilepsy phenotype. The cortex and thalamus, which are known to participate in absence epilepsy, were involved, but the hippocampus was not. Our results indicate that disruption of the mGluR7a-PICK1 complex is sufficient to induce absence epilepsy–like seizures in rats and mice, thus providing, to the best of our knowledge, the first animal model of metabotropic glutamate receptor–PDZ protein interaction in absence epilepsy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Crunelli, V. & Leresche, N. Childhood absence epilepsy: genes, channels, neurons and networks. Nat. Rev. Neurosci. 3, 371–382 (2002).
Bradley, S.R., Rees, H.D., Yi, H., Levey, A.I. & Conn, P.J. Distribution and developmental regulation of metabotropic glutamate receptor 7a in rat brain. J. Neurochem. 71, 636–645 (1998).
Kinoshita, A., Shigemoto, R., Ohishi, H., van der Putten, H. & Mizuno, N. Immunohistochemical localization of metabotropic glutamate receptors, mGluR7a and mGluR7b, in the central nervous system of the adult rat and mouse: a light and electron microscopic study. J. Comp. Neurol. 393, 332–352 (1998).
Folbergrova, J., Haugvicova, R. & Mares, P. Seizures induced by homocysteic acid in immature rats are prevented by group III metabotropic glutamate receptor agonist (R,S)-4-phosphonophenylglycine. Exp. Neurol. 180, 46–54 (2003).
Gasparini, F. (R,S)-4-phosphonophenylglycine, a potent and selective group III metabotropic glutamate receptor agonist, is anticonvulsive and neuroprotective in vivo. J. Pharmacol. Exp. Ther. 289, 1678–1687 (1999).
Ghauri, M., Chapman, A.G. & Meldrum, B.S. Convulsant and anticonvulsant actions of agonists and antagonists of group III mGluRs. Neuroreport 7, 1469–1474 (1996).
Sansig, G. et al. Increased seizure susceptibility in mice lacking metabotropic glutamate receptor 7. J. Neurosci. 21, 8734–8745 (2001).
Perroy, J. et al. Selective blockade of P/Q-type calcium channels by the metabotropic glutamate receptor type 7 involves a phospholipase C pathway in neurons. J. Neurosci. 20, 7896–7904 (2000).
Lafon-Cazal, M. et al. mGluR7-like metabotropic glutamate receptors inhibit NMDA-mediated excitotoxicity in cultured mouse cerebellar granule neurons. Eur. J. Neurosci. 11, 663–672 (1999a).
Pelkey, K.A., Lavezzari, G., Racca, C., Roche, K.W. & McBain, C.J. mGluR7 is a metaplastic switch controlling bidirectional plasticity of feedforward inhibition. Neuron 46, 89–102 (2005).
Okamoto, N. et al. Molecular characterization of a new metabotropic glutamate receptor mGluR7 coupled to inhibitory cyclic AMP signal transduction. J. Biol. Chem. 269, 1231–1236 (1994).
El Far, O. & Betz, H. G-protein-coupled receptors for neurotransmitter amino acids: C-terminal tails, crowded signalosomes. Biochem. J. 365, 329–336 (2002).
Bertaso, F. et al. MacMARCKS interacts with the metabotropic glutamate receptor type 7 and modulates G protein–mediated constitutive inhibition of calcium channels. J. Neurochem. 99, 288–298 (2006).
O'Connor, V. et al. Calmodulin dependence of presynaptic metabotropic glutamate receptor signaling. Science 286, 1180–1184 (1999).
Dev, K.K. et al. PICK1 interacts with and regulates PKC phosphorylation of mGluR7. J. Neurosci. 20, 7252–7257 (2000).
Hirbec, H. et al. The PDZ proteins PICK1, GRIP, and syntenin bind multiple glutamate receptor subtypes. Analysis of PDZ binding motifs. J. Biol. Chem. 277, 15221–15224 (2002).
Boudin, H. et al. Presynaptic clustering of mGluR7a requires the PICK1 PDZ domain binding site. Neuron 28, 485–497 (2000).
Perroy, J. et al. PICK1 is required for the control of synaptic transmission by the mGluR7 receptor. EMBO J. 21, 2990–2999 (2002).
Burette, A., Wyszynski, M., Valtschanoff, J.G., Sheng, M. & Weinberg, R.J. Characterization of glutamate receptor interacting protein-immunopositive neurons in cerebellum and cerebral cortex of the albino rat. J. Comp. Neurol. 411, 601–612 (1999).
Dev, K.K. Making protein interactions druggable: targeting PDZ domains. Nat. Rev. Drug Discov. 3, 1047–1056 (2004).
Dietz, G.P. & Bahr, M. Peptide-enhanced cellular internalization of proteins in neuroscience. Brain Res. Bull. 68, 103–114 (2005).
Danober, L., Deransart, C., Depaulis, A., Vergnes, M. & Marescaux, C. Pathophysiological mechanisms of genetic absence epilepsy in the rat. Prog. Neurobiol. 55, 27–57 (1998).
van Luijtelaar, E.L. & Coenen, A.M. Two types of electrocortical paroxysms in an inbred strain of rats. Neurosci. Lett. 70, 393–397 (1986).
Frankel, W.N. et al. Development of a new genetic model for absence epilepsy: spike-wave seizures in C3H/He and backcross mice. J. Neurosci. 25, 3452–3458 (2005).
Pietrobon, D. Function and dysfunction of synaptic calcium channels: insights from mouse models. Curr. Opin. Neurobiol. 15, 257–265 (2005).
Meeren, H., van Luijtelaar, G., Lopes da Silva, F. & Coenen, A. Evolving concepts on the pathophysiology of absence seizures: the cortical focus theory. Arch. Neurol. 62, 371–376 (2005).
von Krosigk, M., Bal, T. & McCormick, D.A. Cellular mechanisms of a synchronized oscillation in the thalamus. Science 261, 361–364 (1993).
Xu, J. & Xia, J. Structure and function of PICK1. Neurosignals 15, 190–201 (2006–07).
El Far, O. et al. Interaction of the C-terminal tail region of the metabotropic glutamate receptor 7 with the protein kinase C substrate PICK1. Eur. J. Neurosci. 12, 4215–4221 (2000).
Daw, M.I. et al. PDZ proteins interacting with C-terminal GluR2/3 are involved in a PKC-dependent regulation of AMPA receptors at hippocampal synapses. Neuron 28, 873–886 (2000).
Gardner, S.M. et al. Calcium-permeable AMPA receptor plasticity is mediated by subunit-specific interactions with PICK1 and NSF. Neuron 45, 903–915 (2005).
Cao, G. et al. In vivo delivery of a Bcl-xL fusion protein containing the TAT protein transduction domain protects against ischemic brain injury and neuronal apoptosis. J. Neurosci. 22, 5423–5431 (2002).
Aarts, M. et al. Treatment of ischemic brain damage by perturbing NMDA receptor–PSD-95 protein interactions. Science 298, 846–850 (2002).
Hu, R.Q. et al. γ-hydroxybutyric acid-induced absence seizures in GluR2 null mutant mice. Brain Res. 897, 27–35 (2001).
Avanzini, G., de Curtis, M., Franceschetti, S., Sancini, G. & Spreafico, R. Cortical versus thalamic mechanisms underlying spike and wave discharges in GAERS. Epilepsy Res. 26, 37–44 (1996).
Ramakers, G.M., Peeters, B.W., Vossen, J.M. & Coenen, A.M. CNQX, a new non-NMDA receptor antagonist, reduces spike wave discharges in the WAG/Rij rat model of absence epilepsy. Epilepsy Res. 9, 127–131 (1991).
Chen, L. et al. Stargazin regulates synaptic targeting of AMPA receptors by two distinct mechanisms. Nature 408, 936–943 (2000).
Noebels, J.L., Qiao, X., Bronson, R.T., Spencer, C. & Davisson, M.T. Stargazer: a new neurological mutant on chromosome 15 in the mouse with prolonged cortical seizures. Epilepsy Res. 7, 129–135 (1990).
Letts, V.A. et al. The mouse stargazer gene encodes a neuronal Ca2+-channel γ subunit. Nat. Genet. 19, 340–347 (1998).
Zhang, Y., Mori, M., Burgess, D.L. & Noebels, J.L. Mutations in high voltage–activated calcium channel genes stimulate low voltage–activated currents in mouse thalamic relay neurons. J. Neurosci. 22, 6362–6371 (2002).
Andre, V., Pineau, N., Motte, J.E., Marescaux, C. & Nehlig, A. Mapping of neuronal networks underlying generalized seizures induced by increasing doses of pentylenetetrazol in the immature and adult rat: a c-Fos immunohistochemical study. Eur. J. Neurosci. 10, 2094–2106 (1998).
Dalezios, Y., Lujan, R., Shigemoto, R., Roberts, J.D. & Somogyi, P. Enrichment of mGluR7a in the presynaptic active zones of GABAergic and non-GABAergic terminals on interneurons in the rat somatosensory cortex. Cereb. Cortex 12, 961–974 (2002).
Somogyi, P. et al. High level of mGluR7 in the presynaptic active zones of select populations of GABAergic terminals innervating interneurons in the rat hippocampus. Eur. J. Neurosci. 17, 2503–2520 (2003).
Wan, H. & Cahusac, P.M. The effects of L-AP4 and L-serine-O-phosphate on inhibition in primary somatosensory cortex of the adult rat in vivo. Neuropharmacology 34, 1053–1062 (1995).
Turner, J.P. & Salt, T.E. Group III metabotropic glutamate receptors control corticothalamic synaptic transmission in the rat thalamus in vitro. J. Physiol. (Lond.) 519, 481–491 (1999).
Alexander, G.M. & Godwin, D.W. Metabotropic glutamate receptors as a strategic target for the treatment of epilepsy. Epilepsy Res. 71, 1–22 (2006).
Snead, O.C. III, Banerjee, P.K., Burnham, M. & Hampson, D. Modulation of absence seizures by the GABA(A) receptor: a critical role for metabotropic glutamate receptor 4 (mGluR4). J. Neurosci. 20, 6218–6224 (2000).
Steinberg, J.P. et al. Targeted in vivo mutations of the AMPA receptor subunit GluR2 and its interacting protein PICK1 eliminate cerebellar long-term depression. Neuron 49, 845–860 (2006).
Paxinos, G., Watson, C., Pennisi, M. & Topple, A. Bregma, lambda and the interaural midpoint in stereotaxic surgery with rats of different sex, strain and weight. J. Neurosci. Methods 13, 139–143 (1985).
Kim, D. et al. Lack of the burst firing of thalamocortical relay neurons and resistance to absence seizures in mice lacking α(1G) T-type Ca2+ channels. Neuron 31, 35–45 (2001).
Acknowledgements
We thank A. Cohen-Solal for animal handling, M.-C. Rousset and M. Gien-Asari for assistance with immunohistochemistry and movie preparation, and A. Depaulis, B. Chanrion and C. Sharpe for helpful discussion. This work was supported by the Agence Nationale de la Recherche (ANR-05-NEURO-035 and ANR-06-NEURO-035), the Max-Planck-Gesellschaft, European Community (QLG3-CT-2001-00929), the Fonds der Chemischen Industrie and Ligue Française Contre l'Epilepsie (F.B.).
Author information
Authors and Affiliations
Contributions
F.B. and M.L.-N. conducted the experiments, F.d.B. and P.F. handled the softwares for data analyses, J.B. and L.F. supervised the project, P.M. contributed to the design of the TAT peptides, C.Z., A.S. and H.B. generated the mGluR7AAA/AAA mouse and R.L.H. generated the Pick1−/− mouse. F.B., M.L.-N., J.B., L.F., C.Z., A.S. and H.B. wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Methods (PDF 1536 kb)
Supplementary Movie 1
Movie and simultaneous EEG recording obtained 30 min after injection with 1 μmol TAT–R7-LVI (front rat) or TAT–R7-AAA (back rat). Note locomotor arrest and facial myoclonus in the front rat, as compared with normal exploratory activity of the control rat. (MOV 1135 kb)
Supplementary Movie 2
Movie and simultaneous EEG recording of absence-like behavior of an mGluR7aAAA/AAA mouse. Note a similar facial myoclonus and locomotor arrest as in TAT–R7-LVI–injected rat of Movie 1. (MOV 958 kb)
Rights and permissions
About this article
Cite this article
Bertaso, F., Zhang, C., Scheschonka, A. et al. PICK1 uncoupling from mGluR7a causes absence-like seizures. Nat Neurosci 11, 940–948 (2008). https://doi.org/10.1038/nn.2142
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2142
This article is cited by
-
Neuroligin 2 regulates absence seizures and behavioral arrests through GABAergic transmission within the thalamocortical circuitry
Nature Communications (2020)
-
Cellular and pathophysiological consequences of Arp2/3 complex inhibition: role of inhibitory proteins and pharmacological compounds
Cellular and Molecular Life Sciences (2019)
-
Nav1.2 haplodeficiency in excitatory neurons causes absence-like seizures in mice
Communications Biology (2018)
-
Structure function relations in PDZ-domain-containing proteins: Implications for protein networks in cellular signalling
Journal of Biosciences (2018)
-
Elfn1 recruits presynaptic mGluR7 in trans and its loss results in seizures
Nature Communications (2014)